Living With A Stimulator
Once the DBS has been programmed, you are sent home with instructions for adjusting your own stimulation. The handheld controller allows you turn the stimulator on and off, select programs, and adjust the strength of the stimulation. Most patients keep their DBS system turned on 24 hours day and night. Some patients with essential tremor can use it during the day and turn off the system before bedtime. Your doctor may alter the settings on follow-up visits if necessary.
If your DBS has a rechargeable battery, you will need to use a charging unit. On average charging time is 1 to 2 hours per week. You will have a choice of either a primary cell battery or a rechargeable unit and you should discuss this with you surgeon prior to surgery.
Just like a cardiac pacemaker, other devices such as cellular phones, pagers, microwaves, security doors, and anti theft sensors will not affect your stimulator. Be sure to carry your Implanted Device Identification card when flying, since the device is detected at airport security gates.
Peak Calling And Read Counting
Peak calling procedure was carried out in two complementary approaches: 1) Per group : the peaks are called separately for each group, aggregating the relevant samples and inputs. While this approach is not optimal for differential peak calling analysis as it requires harmonization of peaks between the two groups, it can be used to compare the total genome coverage as well as unique H3K27ac genomic regions in each group. 2) All samples combined: the peaks are called by aggregating all samples and input controls. This approach does not allow identification of unique genomic regions however, it provides a better identification of consensus peaks, and thus, was used for differential peak analysis. The resulting peak-sets were filtered using the ENCODE black listed regions and only peaks within canonical chromosomes were kept for downstream analyses. Sample-specific enrichment on the identified peaks was performed using featureCounts version 1.6.4 with default parameters. For both the individual group and all samples combined approaches, peaks with non-adjusted p-value > 107 were excluded from the analysis. Reads inside peaks were quantified using featureCounts program from the subread package v2.0.0. Additional details are provided in the , methods section.
And So Will Be The Mea
Using this kind of 3D structure has implications for the MEA, which is still 2D. Imec is working on a 3D MEA by stacking several MEAs on top of each other. Moreover, they will remove most of the silicon, forming a mesh with holes and nodes. That way, the cells in the organoids can grow through the openings, establishing contact with the electrodes in three dimensions. The goal is to maintain the resolution that the 2D MEA already offers even though mm-size organoids contain far more cells to record from. It is a challenge that researchers already faced in the development of neuroprobes. Now, with the high-density Neuropixels brain implant, we can record signals from many cells in a specific brain volume. The difference is that when implanting a neuroprobe, the pre-existing brain is the limit, and moving to another brain area in a circuit often requires a second device. With the MEA, we can build our own circuits around the electrodes.
A future vision with 3D organoids on a 3D MEA.
Read Also: Pesticides And Parkinson Disease A Critical Review
Awake Vs Asleep Surgery
Standard DBS surgery is performed while you are awake and requires that you stop taking the medicines that control your Parkinson’s symptoms. During surgery, you are asked to perform tasks to help guide the electrode to the precise location in the brain.
Being awake during brain surgery, or being off medicine, is unsettling for some people. Asleep DBS is an alternative option at some centers.
Asleep DBS surgery is performed while you are unconscious and under anesthesia. Surgery takes place in an MRI or CT scanner to target and verify accurate placement of your DBS electrodes. Ask your surgeon if asleep DBS is an option for you.
| Must hold medications the morning of surgery | Don’t have to hold medications |
The Symptoms That Dbs Treats
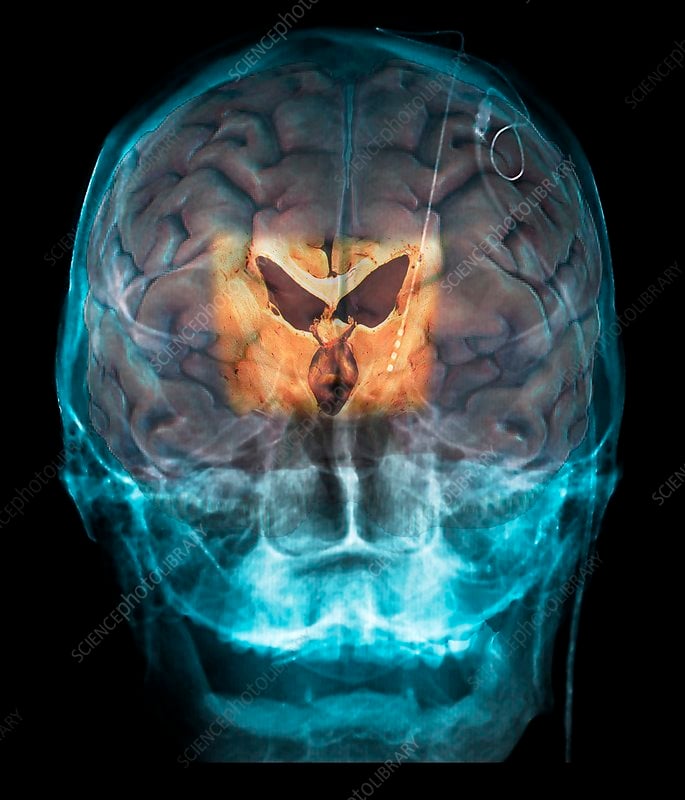
Deep brain stimulation is used primarily to treat the motor symptoms of Parkinsons disease, but this can vary somewhat between the different placement sites. Symptoms treated include:
- Stiffness
- Abnormal movements : Dyskinesias are often a side effect of medications for Parkinsons disease and include involuntary movements such as twisting, head bobbing, squirming, and more.
DBS is not usually helpful with walking problems or balance, though improvements in the symptoms above can indirectly affect walking. It also does not provide significant benefits for non-motor symptoms of Parkinsons such as cognitive changes, mood changes , or problems with sleeping.
The benefits of DBS can be estimated by looking at how a person responds to levodopa. Symptoms that respond to levodopa will often respond to DBS . But symptoms that are not changed with levodopa are unlikely to be improved by DBS.
DBS often allows for a reduction in the dosage of levodopa, which in turn can result in fewer involuntary movements and a reduction in off time. The result is often improved quality of life.
Also Check: Gift Ideas For Parkinson Patients
How Chip Technology Will Decipher Brain Diseases
Imecs chip technology instrumental is decoding brain diseases. By growing relevant circuits on chip, the mechanisms behind Parkinsons can be unraveled
Mission Lucidity brings clinicians, scientists, and engineers from imec, KU Leuven, UZ Leuven, and VIB together to gain a deeper understanding of neurodegenerative disorders with the ultimate goal of curing and preventing them. One of the projects tackles Parkinsons disease. Central in this endeavor is imecs high-density multi-electrode array that can interface at a single-cell level. With stem cell technology that can reprogram cells, the MEA can create and tweak any relevant brain circuit to study disease mechanisms and the effects of drugs on the circuits. Currently, the first patient-derived Parkinsons brain circuits have been grown successfully on the MEA.
Microfluidics Based Brain Cancer Chips
Recapitulating simple organs in vitro is difficult, modeling tumors, cancer progression, extravasations, and metastases are way worse. The tumor highly interacts with its microenvironment that makes spheroid, transwell cell cultures, and organoid unsuitable for mimicking a cancerous tissue. The associated tumor intestinal fluid pressure and mechanical/chemical transfer with the surrounding cells and the local tissue add to the complexity of the pathophysiology of the disease. A reliable in vitro microfluidic model can be of significant help in discovering new anticancer therapies as well as monitoring the patients progress in response to the drug. Brain Chips exploit microfluidic technology to provide a human-relevant microenvironment for studying tumors by offering control over the fluid flow, biochemical and mechanical cues, etc. Examples of microfluidic brain cancer chips are as follows:
Tumor growth modeling: These microfluidics chips are excellent tools for understanding the mechanisms by which the tumor influences the neighboring cells as well as how it gets affected.
Angiogenesis: Angiogenesis is associated with formation and sprouting of new blood vessels and is the hallmark of brain tumors especially glioblastomas, the most malignant brain tumor. Various microfluidic chips have been tested so far for angiogenesis recapitulation. They are normally lined with an ECM gel followed by introducing endothelial cell and monitored for capillary network formation.
Read Also: Alan Alda Parkinson’s Diagnosis
Efficacy Of Segmented Versus Conventional Electrodes
VTA can be modified according to requirement by using segmented electrodes instead of cylindrical ones, as this allows field steering and independent control over electrode contacts.9,60,61 The segmented electrodes have a better design, lesser side effects, and cause lesser activation of surrounding tissue than cylindrical ones hence they provide a larger therapeutic window and give the time to clinicians to modify side-effect thresholds.9,60,61
Summary And Future Prospects
The review shows that DBS is an effective and evolving treatment strategy in PD. However, despite its benefits, it is not widely accepted or widely available due to cost and need for highly skilled staff to manage the programming and stimulation. Additionally, there are challenges of repeat surgeries and short battery life. DBS efficacy, accuracy and acceptability can be improved by overcoming these challenges.
Therefore, upcoming DBS technologies would need to focus on reducing electrode contact size and stimulation number, and move from an invasive to a non-invasive approach.9,67 Newer technologies like current steering can reduce the VTA.67 The VANTAGE study shows that multiple-source axially asymmetric directional DBS using segmented electrodes can be more efficacious than cDBS technologies without steering.60,61 Newer biomarkers and electrode designs like rhythmic neural activity and evoked potentials for segmented electrodes can reduce the programming time.9 Automated support tools to determine the optimal stimulation parameters can increase the penetration of technology in areas where highly skilled staff are not available.9
Today updated software using imaging technologies allows patient specific reconstruction of DBS leads with fully automated tools such as PaCER.67 Recently, a smartphone based wearable and wireless accelerometer and gyroscope platform was successfully used for quantified feedback for optimal configuration of CL-DBS tuning parameters.68
Don’t Miss: Drugs For Parkinson’s Dementia
Will I Have To Limit My Activity Following Deep Brain Stimulation Surgery
- You should not engage in light activities for 2 weeks after surgery. This includes housework and sexual activity.
- You should not engage in heavy activities for 4 to 6 weeks after surgery. This includes jogging, swimming, or any physical education classes. Anything strenuous should be avoided to allow your surgical wound to heal properly. If you have any questions about activities, call your doctor before performing them.
- You should not lift more than 5 lbs. for at least 2 weeks.
- You should not raise your arms above your shoulders or over bend or stretch your neck.
- Depending on the type of work you do, you may return to work within 4 to 6 weeks.
Treating Parkinsons With Medication Dbs And Exercise
Because Parkinsons cannot be cured, all treatments aim to reduce the severity of the symptoms and improve the patients quality of life. Typically, this entails a combination of drugs, increased aerobic exercise and Deep Brain Stimulation .
Parkinsons is caused by a loss of dopamine in the brain, which means that most drugs used to treat it are either converted into dopamine in the brain or imitate the action of dopamine.
Common types of Parkinsons drugs include levodopa and dopamine agonists. Sometimes they are combined with other drugs, such as carbidopa and benzerazide to improve the drugs effectiveness and reduce side effects.
There can be considerable side effects, however. These include:
- Tiredness
- Obsessions with food, gambling, shopping and Internet use
- Some drugs may also lose their effectiveness over time
The surgery for Deep Brain Stimulation involves placing electrodes into areas of the brain that control movement. A pacemaker-type device called a neurostimulator is then typically placed under the skin below the collarbone.
It then sends constant electrical pulses into the brain, thereby decreasing the symptoms of Parkinsons. Doctors will monitor the settings and medication over time, to find the perfect balance of drugs and electrical pulses. Finding this balance can take up to a year. After DBS, patients can often reduce their medication.
You May Like: What Are Off Periods In Parkinson’s
Testing Before Deep Brain Stimulation
For patients with Parkinsons disease, the doctor must confirm that the PD is levodopa-responsive and determine which symptoms are most likely to respond to DBS and discuss these with the patient.
To accomplish these two objectives, the movement disorders neurologist will examine the patient in the absence of his or her PD medications, then again after having taken them. Seeing the effect of PD medications on the movement and non-motor symptoms helps the physician and patient identify good target symptoms for DBS.
A cognitive assessment can help determine a persons ability to participate in the procedure, which involves providing feedback to the doctor during surgery and throughout the neurostimulator adjustment process. This assessment also informs the team of the risk of having worsened confusion or cognitive problems following the procedure.
Some hospitals also perform an occupational therapy review or speech, language and swallowing assessment. A psychiatrist may examine the person to determine if a condition such as depression or anxiety requires treatment before the DBS procedure.
Development Of Organoid Models To Study Pd

Fig. 2: Present and future in vitro models for PD.
a Existing approaches to model PD using iPSC-derived neurons/brain organoids. b Future prospect can combine several organoids to make an assembloid system where microbiota derived metabolites could be injected and thus it can mimic the gut brain axis in PD. c Further usage of the organ on chip system could mimic the gut brain axis where the sophisticated fluidics system can establish a connection between two on chip organs. In one chip it is possible to create intestine and brain in the other. The microfuidic system would establish the connection between the two and there would be an injection site for bacterial metabolites as depicted in the image. These approaches will improve the PD modeling and can be used to study disease progression or it can help in bulk drug screening.
Also Check: Treatment For Parkinson’s Disease Usually Includes
Data Management Design Inputs
Having the object of accelerating neurophysiological research, a core requirement for a bidirectional IPG acting as a clinical BCI is to store and transmit neural signals. Although chronic data streaming represents a heuristic goal, its practical implementation still needs to overcome important limitations such as high-power demand, consequent fast battery drain, and maintenance of a permanent external receiver link all these features ultimately add unnecessary burdens for patients. For instance, continuous data streaming with an implantable rechargeable device require the use of a transmitter that has to be continuously worn by the patient. Many bidirectional neuromodulation platforms are targeting chronic wireless communication at the preclinical or investigational stage.
Embedding compressed data requires to have an a priori knowledge of what signal features are significant for the specific disease, but because of the exploratory application of clinical BCI, time domain data are necessary to the discovery of new biomarkers and physiological mechanisms of action. Moving from the concept of chronic monitoring to exploratory recording, the requirement of data wireless streaming can be relaxed by limiting it to on-demand and time-constrained streaming sessions that allow for controlled experimental investigations without burdening the patient.
How Is Parkinsons Disease Diagnosed
While there are no specific tests available that diagnose Parkinsons, when symptoms start to show, doctors will often refer patients to a neurologist.
These specialists will diagnose Parkinsons by studying the patients medical history and the symptoms that are showing. A physical and neurological examination will also take place and sometimes a dopamine transporter scan.
A number of tests like blood work along with MRIs, CT scans and ultrasounds of the brain may be ordered to rule out other possible illnesses. It can take time to diagnose Parkinsons and doctors may sometimes prescribe Parkinsons medication to confirm the diagnosis. If the medication brings about considerable improvement in the symptoms, then this will confirm the diagnosis of Parkinsons.
Don’t Miss: What Is The Last Stage Of Parkinson Disease
Concerns And Ethical Considerations
Ethical questions raised include who are good candidates to receive neural implants and what are good and bad uses of neural implants.Whilst deep brain stimulation is increasingly becoming routine for patients with Parkinson’s disease, there may be some behavioural side effects. Reports in the literature describe the possibility of apathy, hallucinations, compulsive gambling, hypersexuality, cognitive dysfunction, and depression. However, these may be temporary and related to correct placement and calibration of the stimulator and so are potentially reversible.
Some transhumanists, such as Raymond Kurzweil and Kevin Warwick, see brain implants as part of a next step for humans in progress and evolution, whereas others, especially bioconservatives, view them as unnatural, with humankind losing essential human qualities. It raises controversy similar to other forms of human enhancement. For instance, it is argued that implants would technically change people into cybernetic organisms . It’s also expected that all research will comply to the . Yet further, the usual legal duties apply such as information to the person wearing implants and that the implants are voluntary, with few exceptions.
Other concerns involve vulnerabilities of neural implants to cybercrime or intrusive surveillance as neural implants could be hacked, misused or misdesigned.
Minimizing Dbs Side Effects For Improved Efficacy
Efficacy of DBS will be higher if the safety profile is manageable and the procedure is acceptable to the patients. Since it is a surgical procedure, usual surgery associated complications such as hemorrhage, infection, and skin erosion are seen.64 Other than these, hardware-related complications such as electrode fracture and stimulator failure have been reported.65
However, the most annoying, common and important postoperative complication of DBS is the side effects caused by electrical stimulation of the surrounding brain tissue these are largely driven by the DBS target and anatomy and functionality of the surrounding brain tissue.66 However, these side effects are easily manageable in most cases.66 Emerging technologies such as segmented electrodes and CL-DBS aim to minimize these side effects.9
You May Like: Stages Of Parkinson’s What To Expect
How Deep Brain Stimulation Works
Exactly how DBS works is not completely understood, but many experts believe it regulates abnormal electrical signaling patterns in the brain. To control normal movement and other functions, brain cells communicate with each other using electrical signals. In Parkinson’s disease, these signals become irregular and uncoordinated, which leads to motor symptoms. DBS may interrupt the irregular signaling patterns so cells can communicate more smoothly and symptoms lessen.
Programming Cells On A Mea
In a first step fibroblast progenitor cells are harvested from a patients skin and plated on the MEA. The imec MEA consists of 16.384 individually steerable electrodes at single-cell resolution. The electrodes are spaced so close together that each cell is in contact with at least one electrode. By sending a small current through the electrode, tiny transient holes can be made in the cell in a process called electroporation. Transcription factors are then applied on the cells and enter through the holes to reprogram the cell into brain cells.
Because the MEA can access single cells, researchers can build the striatal circuit by choosing which cells are becoming either cortical, striatal, or dopaminergic cells on specific locations of the chip. By selectively activating the electrodes, a variety of patterns can be built to create microcircuits. The MEA fits into a cell culture incubator and connects to a computer with a custom user interface. The idea is that the system will be distributed over different labs to initiate a global partnership.
Single cell electroporation on the high-density MEA is demonstrated by being able to stimulate cells on demand, to create any desired pattern, like a smiley face. Dextran fluorescein of 10,000 molecular weight was added to the solution and only enters electroporated cells. Neural progenitor cells were stained with calcein and stains all cells.
Recommended Reading: Power Up For Parkinson’s