Genetic Principles And Exceptions Thereof In Familial Pd
The majority of PD cases are sporadic, i.e., only about 10% of patients report a positive family history . Out of the six genes unequivocally linked to heritable, monogenic PD, mutations in SNCA , and LRRK2 are responsible for autosomal-dominant PD forms, and mutations in Parkin , PINK1 , DJ-1 , and ATP13A2 are accountable for PD that displays an autosomal recessive mode of inheritance.
In general, the inheritance patterns of human disorders are identified by examining the way the disorders are transmitted in the family of the index patient. Such a pedigree analysis requires a careful assembly of the disease records of the family members over several generations, and if possible, examination and sample collection from affected and unaffected individuals from the pedigree. All of the currently known monogenic PD forms are autosomal , which means that they are linked with regions on autosomes .
Pedigree of a PD family that comprises affected members with and without the LRRK2 p.G2019S mutation. Five mutation carriers are unaffected, showing reduced penetrance, two mutation carriers are affected with dystonia, showing variable expressivity, and one affected family member does not have the p.G2019S mutation in LRRK2. Black symbols â affected individuals white symbols â unaffected individuals half-filled symbols â individuals with dystonia + â mutation carriers.
Which Gene Mutations May Be Related To Parkinson Disease
Neurodevelopment Neuron Cell Differentiation And Survival
SNPs within loci associated with neurodevelopment, neuron cell differentiation and survival recognized as PD susceptibility factors by GWASs are listed in Table 6.
Table 6. SNPs within loci associated with neurodevelopment, neuron cell differentiation and survival recognized as PD susceptibility factors by GWASs.
CCDC62
CCDC62 was found to be related to PD susceptibility in the Nalls et al. studies conducted in 2011 and 2014 pointing out two different intron variants. The gene has a role in cell growth, estrogen receptor transactivation, cyclin D1 expression in prostate cancer cells as well as in other varieties of cancer, since its antibodies are often produced and thus detected . Until recently, it was mainly reported as related to cancer . In order to define the gene’s function more precisely, more functional studies are required.
RIT2
FGF20
FGF20 was proved to be associated with PD in the Nalls et al. meta-analysis , in which one intron variant within the gene with a positive association was found. More studies will have to be done to confirm this association. FGF20 is a neurotrophic factor preferentially expressed in substantia nigra pars compacta. It acts in an autocrine/paracrine manner. FGF20 regulates central nervous development and function . It plays a major role in dopaminergic neurons differentiation and survival . According to some studies, FGF20 also increases SNCA levels in dopaminergic neurons, but there is a huge discrepancy among studies .
GCH1
GPNMB
Genetic Predispositions Reducing Differentiation Yield Of Mda Neurons
In vitro neural development was impaired in neural lines derived from patients carrying LRRK2, PRKN, SNCA, and sporadic mutations,,,. In four independent studies, the differentiation potential of neural progenitor cells derived from patients was significantly reduced, demonstrated by low yields of neurons in comparison with control lines,,,,. A recent review presented the idea that PD is attributed to significant neurodevelopmental defects, which may increase the susceptibility for disease onset. If confirmed, identifying genetic predispositions that contribute to early developmental defects in iPSC-PD may assist the development of novel PD therapies. However, these phenotypes may appear in conflict with other studies,, capable of generating functional neurons from cell lines with similar mutations. The differences could be due to varying protocols, which may be more or less stressful for the cells.
Recommended Reading: What Is The Difference Between Alzheimer’s And Parkinson’s Disease
What Is Parkinsons Disease
Parkinsons disease is the second most common neurodegenerative disorder and the most common movement disorder. Characteristics of Parkinsons disease are progressive loss of muscle control, which leads to trembling of the limbs and head while at rest, stiffness, slowness, and impaired balance. As symptoms worsen, it may become difficult to walk, talk, and complete simple tasks.
The progression of Parkinsons disease and the degree of impairment vary from person to person. Many people with Parkinsons disease live long productive lives, whereas others become disabled much more quickly. Complications of Parkinsons such as falling-related injuries or pneumonia. However, studies of patent populations with and without Parkinsons Disease suggest the life expectancy for people with the disease is about the same as the general population.
Most people who develop Parkinsons disease are 60 years of age or older. Since overall life expectancy is rising, the number of individuals with Parkinsons disease will increase in the future. Adult-onset Parkinsons disease is most common, but early-onset Parkinsons disease , and juvenile-onset Parkinsons disease can occur.
Genetic Susceptibility Factors In Parkinson’s Disease
Monogenic forms represent less than 10% of PD in most populations. The vast majority result from complex interactions among genes and between genes and environmental factors. Genetic variations may be susceptibility factors or disease modifiers, affecting penetrance, age at onset, severity and progression. High-density arrays of single nucleotide polymorphisms permit the identification of susceptibility factors in genome-wide association studies, in which the frequencies of putative risk alleles are compared in patients and controls.
Read Also: Does Sam Waterston Have Parkinson
How Often Does Parkinsons Run In The Family
Most Parkinsons cases have no connection to a genetic cause, but scientists have found that some gene mutations can heighten an individuals risk. Researchers believe that a better understanding of these genes may improve ways of identifying and treating the illness.
The National Institute of Neurological Disorders and Stroke reports that an estimated 15 to 25 percent of people with Parkinsons have a family history of the disorder. The Michael J. Fox Foundation for Parkinsons Research estimates that about 10 percent of cases are linked with a genetic cause.
Parkinsons doesnt stand out as a hereditary disease over and above any other chronic diseases that people deal with, says Rebecca Gilbert, MD, PhD, chief scientific officer for the American Parkinson Disease Association in New York City. But if you have a parent with Parkinsons disease, you have about a fourfold greater risk over the general population.
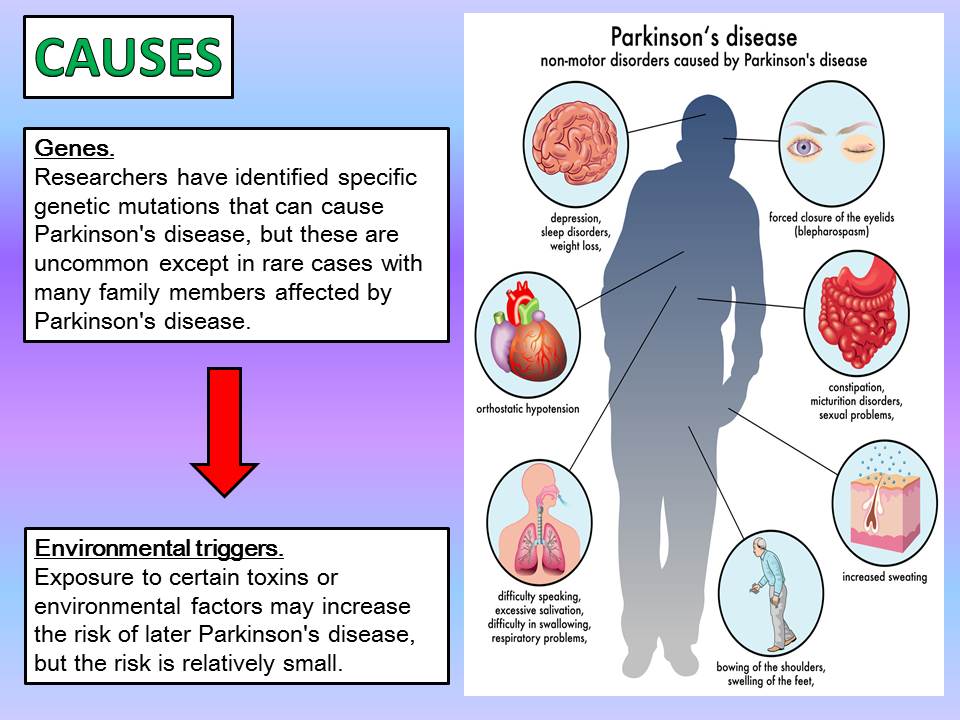
Still, that risk is relatively small. About 1 percent of the population over 60 has Parkinsons, according to the Michael J. Fox Foundation, and that number rises to about 4 percent for those who have a mother or father with the illness, according to Dr. Gilbert. The overall message is: Just because you have a gene linked to Parkinsons does not mean you will get the disease.
When To See A Doctor About Parkinsons
There isnt one specific test to diagnose Parkinsons disease. Doctors will usually evaluate your symptoms and perform several tests to determine if you have the condition. If you notice the following early warning signs, then you should see a doctor.
The early warning signs of Parkinsons disease include:
You May Like: Nursing Management Of Parkinson’s Disease
Genetics Of Parkinson’s Disease Risk
Risk for Parkinson’s disease is influenced by genetics. In fact, the disease is associated with genetic variants in many genes. Two of the best studied genes associated with Parkinson’s disease are called LRRK2 and GBA. There are other genesand variants that have also been linked to Parkinson’s disease, but most of these genetic variants are either rare or have only a smalleffect on risk.
Genetic Testing: Whom To Test And How
The answer to the question of who should be tested is not trivial. To date, no formal testing guidelines have been developed by the Movement Disorder Society or any other PD alliance group. Out of all monogenic forms, mutations in LRRK2, Parkin, and PINK1 are the most likely to be encountered in clinical practice. In the following scenarios, genetic testing might prove useful to minimize further patient workup, to clarify treatment approaches, and/or to assist with future family planning: juvenile-onset PD irrespective of family history early-onset PD with atypical features and/or a positive family history of this disease or late-onset PD with a strong family history of PD . Guidelines published by the European Federation of the Neurological Sciences recommend screening LRRK2 for mutations in Europeans showing dominant inheritance of PD, testing for the LRRK2 p.G2019S mutation in familial and sporadic cases of PD in specific populations, and analysis of Parkin, PINK1, and DJ-1 in patients aged < 35 yr with recessively inherited PD .
Also Check: How Long Does A Parkinson’s Patient Live
The Genetic Architecture Of Parkinsons Disease In Latino Populations
An international research team led by Cleveland Clinic has presented the most comprehensive characterization of the underlying genetic basis for Parkinsonâs disease in Latinos to date, marking an important step towards more inclusive PD genetic research.âParkinsonâs disease impacts all ethnic groups, but since genetic studies have largely been limited to individuals of European and East Asian ancestry, little is known about the genetic architecture of the disease in Latino populations, â said Ignacio Mata, Ph.D., assistant staff in the Genomic Medicine Institute and lead author on the study. âAs we see incidence rates rise in nearly every global region, the importance of greater diversity in Parkinsonâs research cannot be overlooked.âIn this study, published in Annals of Neurology, Dr. Mata and international collaborators performed the first ever genome-wide association study of Latino PD patients from South America. Their analysis relied on patient data from the worldâs largest PD case-control cohort of Latinos, called the Latin American Research Consortium on the Genetics of Parkinsonâs Disease , which includes individuals from 35 institutions in 12 countries across Latin America and the Caribbean.
Human Ipsc Studies Of Pd Highlight Converging Molecular And Cellular Pathways Across Genetic Subgroups
Our analysis of 385 iPSC-derived cell lines from 67 published studies reveals that many PD neuronal phenotypes are shared between genetically heterogeneous familial and sporadic patients . Notably, impairments in mechanisms involved in cellular waste recycling, mitochondrial function, neuronal morphology and physiology, and sensitivity to reactive oxygen species are most common across patient lines with varying genetic predispositions . The studies measured cellular phenotypes that occurred either spontaneously or in response to chemicals mimicking cellular aging and stress . It is important to note that the frequency of reported phenotypes in our meta-analysis may be biased because only few studies reported negative results ,,,,,,,,,,,,. In addition, most cell lines were not systematically phenotyped without prior hypothesis and thus, there is likely to be an ascertain bias in these phenotypes. Less hypothesis-driven multimodal or omics analysis will help to address such bias,,,,,,,,. Phenotypes caused by genomic predispositions allude to crosstalk and impairments in multiple pathways that act collectively to mediate selective degeneration of dopaminergic neurons in the substantia nigra and will be discussed in detail below.
Fig. 4: Phenotypic insights from iPSC studies of Parkinsonâs disease.
Also Check: What Foods Should You Avoid If You Have Parkinson’s Disease
Other Causes Of Parkinsonism
Parkinsonism is the umbrella term used to describe the symptoms of tremors, muscle rigidity and slowness of movement.
Parkinsons disease is the most common type of parkinsonism, but there are also some rarer types where a specific cause can be identified.
These include parkinsonism caused by:
- medication where symptoms develop after taking certain medications, such as some types of antipsychotic medication, and usually improve once the medication is stopped
- other progressive brain conditions such as progressive supranuclear palsy, multiple systems atrophy and corticobasal degeneration
- cerebrovascular disease where a series of small strokes cause several parts of the brain to die
You can read more about parkinsonism on the Parkinsons UK website.
Page last reviewed: 30 April 2019 Next review due: 30 April 2022
When Should A Person Seek Genetic Testing

Genetic testing is available for some genes related to Parkinsons disease, but testing may not provide useful information to individuals.
For one thing, a wide range of genes may play a role, and it is not possible to test them all. A person may also have a relevant feature but not go on to develop Parkinsons disease.
For example, only around 0.7% of people with symptoms of Parkinsons disease have changes in the LRRK2 gene, and around 0.3% have changes in the PRKN gene, according to a 2020 review.
Finding out in advance if a young person has the gene may help them prepare for the future if there is strong evidence of a family history of the condition. However, the results are unlikely to be conclusive and may cause unnecessary anxiety.
Anyone who is interested in genetic testing should discuss the pros and cons with a doctor and consider genetic counseling if they decide to go ahead.
The Parkinsons Foundation notes that testing is often hard to access. It can also be costly, and health insurance may not cover it. Genetic counseling can be an additional cost.
Recommended Reading: Parkinson’s And Boxing Connection
Genetic Classification Of Pd
In the current PD genetics nomenclature, 18 specific chromosomal regions, also called chromosomal locus, are termed PARK , and numbered in chronological order of their identification . In addition to being an incomplete list of known PD-related genes, this classification system, unfortunately, has a number of inconsistencies. It comprises confirmed loci, as well as those for which linkage or association could not be replicated . The causative gene has not yet been identified for all of the loci, nor do all of the identified genes contain causative or disease-determining mutations . Finally, one locus, PARK4, was designated as a novel chromosomal region associated with PD but was later found to be identical with PARK1 . It is noteworthy that some of the loci have been identified by genetic linkage analysis in large families, some based on the known function of the protein product of the gene they contain, yet others have been established by genome-wide association studies performed on a population level. A list of the PARK PD-related genes and loci is given in , along with their clinical classification, inheritance pattern , gene , status , and mode of identification.
Advancing Age And Parkinsons Disease
Age is perhaps the biggest risk factor for the onset of Parkinsons disease. The average age at which people will develop this movement disorder is 60. This is not usually something that affects younger people. The brain ages as people get older.
Even without external factors, cells in the substantia nigra can die on their own as an individual ages, causing symptoms to develop as the person gets older.
Read Also: Can Head Injury Cause Parkinson’s
Other Factors Influencing Parkinson’s Disease Risk
Other factors besides genetics can influence someone’s chances of developing Parkinson’s disease, including:
- Age: The risk of developing Parkinson’s disease increases as a person ages.
- Sex: Males have a higher chance of developing Parkinson’s disease than females.
- Family history: First-degree relatives of an individual with Parkinson’s disease have a higher chance of developing Parkinson’s disease.
- Exposure to certain chemicals increases the risk of developing Parkinson’s disease.
Stages Of Parkinsons Disease
- Stage One
- The person will experience mild symptoms that generally do not interfere with daily activities. They may experience tremors and other movement symptoms on one side of the body and changes in posture, walking, and facial expressions can occur.
Read Also: Symptoms Of Parkinsons In Women
What Is The Outlook For Persons With Parkinsons Disease
Although there is no cure or absolute evidence of ways to prevent Parkinsons disease, scientists are working hard to learn more about the disease and find innovative ways to better manage it, prevent it from progressing and ultimately curing it.
Currently, you and your healthcare teams efforts are focused on medical management of your symptoms along with general health and lifestyle improvement recommendations . By identifying individual symptoms and adjusting the course of action based on changes in symptoms, most people with Parkinsons disease can live fulfilling lives.
The future is hopeful. Some of the research underway includes:
- Using stem cells to produce new neurons, which would produce dopamine.
- Producing a dopamine-producing enzyme that is delivered to a gene in the brain that controls movement.
- Using a naturally occurring human protein glial cell-line derived neurotrophic factor, GDNF to protect dopamine-releasing nerve cells.
Many other investigations are underway too. Much has been learned, much progress has been made and additional discoveries are likely to come.
Who Should Get Genetic Testing
Two groups might consider getting genetic testing, according to Gilbert:
- People with Parkinsons who want to know if they have a mutation they may pass along to their children
- Children and siblings of family members with Parkinsons who want to determine their genetic risk for the disease
Right now its not standard of care for everyone with Parkinsons to get genetic testing, she says. The likelihood that were going to find one of these mutations that is known already is small, and even if you have a mutation associated with Parkinsons, it doesnt mean that youre going to get the disease.
So, at this point, the value of getting tested depends on the individual. Doctors can provide this type of genetic evaluation, or people may turn to direct-to-consumer genetic testing, such as 23andMe. These tests, however, can be limited.
You have to be careful with those panels because theyre not very comprehensive, says Gilbert. They may test for only one or two gene variations.
Currently, 23andMe analyzes DNA from spit samples for a variant in LRRK2 and a variant in the GBA gene associated with the disorder. The company makes it clear that the exam does not diagnose the disease, and there are many other mutations to consider.
Parkinsons patient Paul Cannon, PhD, who works for 23andMe as its Parkinsons research community manager, took the test and found that he had neither of the genetic variations.
Don’t Miss: Nursing Home Care For Parkinson’s Patients