Is An Mri Exam Safe For Parkinson’s Disease
Yes. The MRI exam poses no risk to the average person if appropriate safety guidelines are followed. But, if you have a deep brainstimulator to treat your Parkinson’s disease, talk to your doctor before having an MRI since the stimulator may need to be turned off. It should be evaluated and turned on again after the MRI.
Some conditions may make an MRI exam inadvisable. Others mean that precautions need to be taken. Tell your doctor if you have any of the following conditions:
- Weigh more than 300 pounds
- Not able to lie on back for 30 to 60 minutes
- Claustrophobia
Mri Brain Scans Detect People With Early Parkinson’s
Oxford University researchers have developed a simple and quick MRI technique that offers promise for early diagnosis of Parkinson’s disease.
The new MRI approach can detect people who have early-stage Parkinson’s disease with 85% accuracy, according to research published in Neurology, the medical journal of the American Academy of Neurology.
‘At the moment we have no way to predict who is at risk of Parkinson’s disease in the vast majority of cases,’ says Dr Clare Mackay of the Department of Psychiatry at Oxford University, one of the joint lead researchers. ‘We are excited that this MRI technique might prove to be a good marker for the earliest signs of Parkinson’s. The results are very promising.’
Claire Bale, research communications manager at Parkinson’s UK, which funded the work, explains: ‘This new research takes us one step closer to diagnosing Parkinson’s at a much earlier stage one of the biggest challenges facing research into the condition. By using a new, simple scanning technique the team at Oxford University have been able to study levels of activity in the brain which may suggest that Parkinson’s is present. One person every hour is diagnosed with Parkinson’s in the UK, and we hope that the researchers are able to continue to refine their test so that it can one day be part of clinical practice.’
We think that our MRI test will be relevant for diagnosis of Parkinson’s
Dr Michele Hu
How Parkinsons Disease Is Diagnosed
Parkinsons disease is usually diagnosed clinically, meaning that a physician looks for the presence or absence of the possible symptoms of Parkinsons disease by interviewing the patient and performing a detailed neurologic examination.
While there is presently no definitive test for Parkinsons, it can often be identified by a general neurologist, who is trained to diagnose and treat neurologic disorders. To avoid misdiagnosis, consultation with a movement disorder specialist is recommended. A movement disorder specialist is a physician who has undergone additional, subspecialty training in the diagnosis and treatment of movement disorders, such as Parkinsons, after training in general neurology.
What to expect during your visit with a physician
Typically, a trained physician will only consider the diagnosis of Parkinsons disease if the person being examined has at least two of the core motor symptoms of Parkinsons, including tremor, the characteristic bradykinesia , or rigidity. At the end of your visit, the physician should discuss with you why you may or may not have Parkinsons disease and the level of certainty about the diagnosis. This determination is based on your medical history and examination at this visit.
Brain imaging and other tools to aid diagnosis of Parkinsons
Recommended Reading: Parkinsonism Vs Parkinson’s Disease
What Should I Expect During The Mri
As the MRI scan begins, you will hear the equipment making a variety of banging, clanging and muffled thumping sound that will last for several minutes. None of them are anything other than annoying. Other than the sound, you should experience no unusual sensations during the scanning.
Certain MRI exams require an injection of a contrast material. This helps identify certain anatomic structures on the scan images.
Please feel free to ask questions. Tell the technologist or the doctor if you have any concerns.
Dementia With Lewy Bodies
Dementia with Lewy bodies is responsible for approximately 25% of dementias and belongs to the atypical Parkinson syndromes together with progressive supranuclear palsy and multi-system atrophy . The clinical manifestations can be similar to that of AD or dementia associated with Parkinson’s disease. Patients typically present with one of three symptom complexes: detailed visual hallucinations, Parkinson-like symptoms and fluctuations in alertness and attention.Pathologically, the disease is characterized by the presence of Lewy bodies in various regions of the hippocampal complex, subcortical nuclei and neocortex with a variable number of diffuse amyloid plaques. Cholinesterase inhibitors are currently the treatment of choice for this condition.
The role of imaging is limited in Lewy body dementia.Usually the MR of the brain is normal, including the hippocampus. This finding is important as it enables us to differentiate this disease from Alzheimer’ s disease, the main differential diagnosis. Nuclear imaging can be used to demonstrate an abnormal dopaminergic system
PSP with midbrain atrophy
Don’t Miss: Benzodiazepines And Parkinson’s Disease
Imaging And Differential Diagnosis
The core clinical signs of PD include resting tremor, bradykinesia, rigidity, and postural instability. Most patients also experience nonmotor symptoms such as cognitive and emotional changes , dysautonomia, sleep disorders, and sensory disturbances. Many experience prodromal nonmotor symptoms such as anosmia, depression, constipation, and REM sleep behavior. Clinical subtypes of the disease have been identified, including tremor dominant and postural instability gait difficulty . Atypical features may be clues that there are other etiologies that can be differentiated with imaging studies.1 Structural brain imaging is frequently ordered to investigate these cases. In addition, SPECT imaging with DaT may be useful to confirm central nervous system dopamine signaling deficiency in select cases . On DaT scans, normal radiotracer uptake in the striatum forms 2 crescent-shaped regions of activity, mirrored around the median plane. In contrast, in PD, there is asymmetrically decreased activity in the putamen, often with preserved uptake in the caudate nucleus.2,3 A DaT scan is FDA approved for differentiating essential tremor from PD, and is also frequently useful for differentiating drug-induced parkinsonism from PD.
Cerebrovascular Disease
Corticobasal Degeneration
Multiple System Atrophy
Progressive Supranuclear Palsy
Neoplasms
Neurotoxicity
Normal Pressure Hydrocephalus
Quantitative Assessment Of Regional Cerebral Atrophy
As an indirect method of measuring regional brain atrophy, groups have applied simple quantitative measures of diameters, areas and volumes including ROI-based assessment of various structures on MRI for differential diagnostic purposes . In terms of infratentorial atrophy, several studies have demonstrated that MSA is associated with a relatively greater pontine and MCP atrophy compared to PSP and PD, whereas patients with PSP have a relatively greater midbrain and SCP atrophy compared to MSA and PD .
Midsagittal measurements of brainstem areas reveal decreased midbrain areas in PSP patients compared to non-PSP parkinsonian patients and decreased pontine areas in MSA patients compared to non-MSA parkinsonian patients . As single measurements of these structures have been shown not to adequately distinguish between neurodegenerative parkinsonian disorders, especially MSA and PSP, the ratio between ma/pa-ratio was found to be significantly smaller in patients with PSP compared to other groups and to differentiate better than the single measurement .
Despite many advantages of voxel-based analysis, including its independence from operators due to automated detection, at this time it is not appropriate for routine diagnostic work-up of individual patients since it involves group-wise comparisons . Furthermore, in performing a voxel-based study many methodological options are available and known for pitfalls which are summarized in a comprehensive review .
Don’t Miss: Nocturnal Leg Cramps Parkinson’s
Further Testing In Parkinson’s
In other situations, where perhaps the diagnosis is not as clear, younger individuals are affected, or there are atypical symptoms such as tremor affecting both hands or perhaps no tremor at all, further testing may help. For example, imaging can play a role in differentiating between essential tremor and Parkinsons. It can also be important to confirm what is initially a clinical diagnosis of Parkinsons prior to an invasive treatment procedure such as surgical DBS
Functional Imaging And Atypical Pd
Imaging presynaptic dopaminergic terminal function with either striatal 18F-dopa uptake or a DAT SPECT marker shows high sensitivity for detecting atypical parkinsonian syndromes but only poor specificity for discriminating them from typical PD . The typical gradient of loss of dopaminergic function in PD, where the head of the caudate is relatively spared, is less evident in progressive supranuclear palsy and corticobasal degeneration patients . In progressive supranuclear palsy, there is a more symmetric pattern of nigrostriatal dysfunction than in other parkinsonian syndromes.
In contrast, measurements of resting glucose metabolism can be helpful for separating typical from atypical parkinsonian syndromes. In typical idiopathic PD, lentiform nucleus glucose metabolism is preserved or raised, whereas it is reduced in most atypical cases .
FIGURE 5.
18F-FDG PET images of PD and multiple-system atrophy patient. Multiple-system atrophy patient shows significant striatal reduction of glucose metabolism. MSA = multiple-system atrophy.
Read Also: Claiming Pip For Parkinson’s Disease
Regional Cerebral Glucose Metabolism And Pd
18F-FDG PET can be used to assess levels of resting regional cerebral glucose metabolism. Absolute levels in the lentiform nucleus lie within the reference range in PD however, covariance analysis reveals an abnormal profile of relatively raised resting lentiform nucleus and lowered frontal and parietotemporal metabolism . The degree of expression of this PD-related profile correlates with clinical disease severity, thus providing a potential biomarker of disease progression. Successful treatment with levodopa or deep brain stimulation reduces expression of the PD-related profile . Given this, changes in treatment could result in a potential confounding factor if 18F-FDG PET were to be used as a biomarker to follow PD progression.
Eckert et al. performed 18F-FDG PET on 8 patients with suspected early parkinsonism but normal findings on 18F-dopa PET . None of these 8 patients expressed a PD-related profile of glucose metabolism, and over 3 y none of them showed any clinical progression of their disorder. This finding reinforces the viewpoint that normal dopaminergic imaging excludes the presence of a degenerative parkinsonian syndrome.
Testing For Parkinsons Disease
There is no lab or imaging test that is recommended or definitive for Parkinsons disease. However, in 2011, the U.S. Food and Drug Administration approved an imaging scan called the DaTscan. This technique allows doctors to see detailed pictures of the brains dopamine system.
A DaTscan involves an injection of a small amount of a radioactive drug and a machine called a single-photon emission computed tomography scanner, similar to an MRI.
The drug binds to dopamine transmitters in the brain, showing where in the brain dopaminergic neurons are.
The results of a DaTscan cant show that you have Parkinsons, but they can help your doctor confirm a diagnosis or rule out a Parkinsons mimic.
Don’t Miss: When Was Parkinson’s Disease Discovered
Exclusion Of Alternative Diagnoses
Structural MRI with conventional MR sequences is usually normal in early PD patients limiting its application in clinical routine for the detection of early PD. Recent studies, however, identified imaging correlates of underlying neuropathology in PD patients through advanced MRI techniques. These imaging abnormalities will be discussed in detail later in this review. Nevertheless, cMRI was repetitively shown to be useful in discriminating PD from APDs such as MSA and PSP. Latter are characterized by disease-specific atrophy patterns and signal intensity changes. In addition, current operational diagnostic criteria require the exclusion of symptomatic causes of parkinsonism in the work-up of patients with PD .
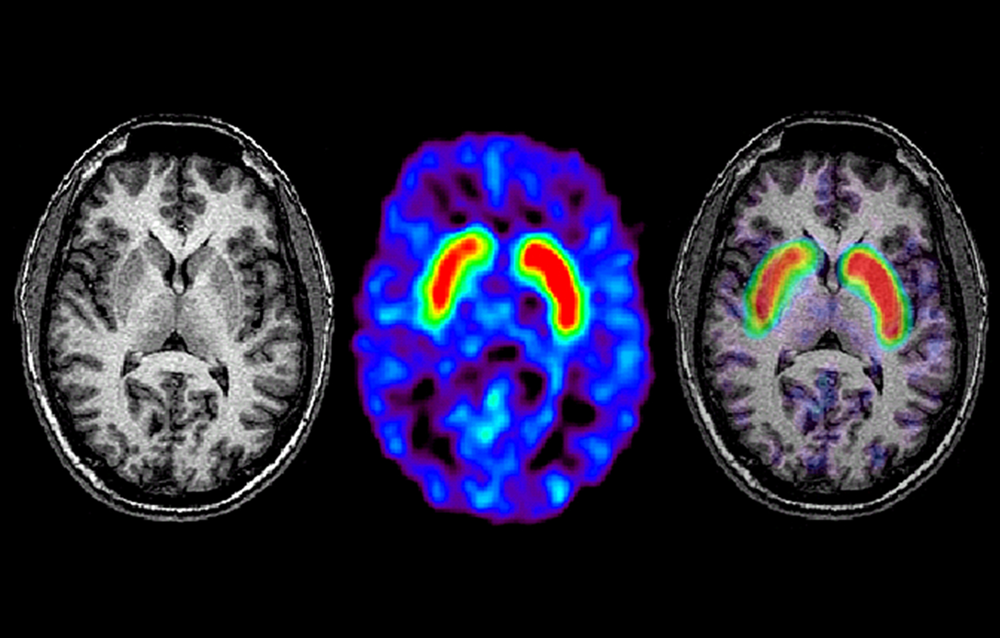
Imaging Presynaptic Dopaminergic Function In Pd
The function of dopamine terminals in PD can be examined in vivo in 3 main ways . First, the availability of presynaptic dopamine transporters can be assessed with a variety of PET and SPECT tracers, most of which are tropane-based. Examples include 123I–2-carbomethoxy-3-tropane , 123I-N-3-fluoropropyl-2-carbomethoxy-3-tropane , 123I-altropane, and 11C-2-carbomethoxy-3-tropane . Second, 18F-3,4-dihydroxyphenylalanine PET provides a marker of terminal dopa decarboxylase activity and dopamine turnover. Third, vesicle monoamine transporter availability in dopamine terminals can be examined with either 11C- or 18F-dihydrotetrabenazine PET. Early hemiparkinsonian patients show bilaterally reduced putamen dopaminergic terminal function, activity being more depressed in the posterior putamen contralateral to the affected limbs . Head-of-caudate and ventral striatal function is relatively preserved. Clinical parkinsonism occurs when PD patients have lost 40%50% of posterior putamen dopamine terminal function . Levels of putamen 18F-dopa uptake and DAT binding correlate inversely with bradykinesia and rigidity of PD patients, but interestingly, not with tremor severity . This suggests that parkinsonian tremor is not a direct consequence of nigrostriatal degeneration.
FIGURE 4.
123I-FP-CIT SPECT images of healthy volunteer and patient with early hemi-PD. PD patient shows asymmetric bilateral loss of putamen DAT binding.
Also Check: Overactive Thyroid And Parkinson’s
T Mri Measures Changes In Brains Of Patients With Parkinsons Disease
Researchers at the University of Cambridge used a new ultra-high strength 7T MRI scanner at the Wolfson Brain Imaging Centre to measure changes in the brains of people with Parkinsons disease, progressive supranuclear palsy , or in good health. The study, published in Movement Disorders, suggests that 7T MRI scanners could be used to help identify those patients with Parkinsons disease and similar conditions most likely to benefit from new treatments for previously untreatable symptoms.
Patients with Parkinsons disease and PSP are often treated with drugs such as L-DOPA, which compensate for the severe loss of dopamine. But, dopamine treatment does little for many of the non-motor symptoms. That is why scientists have begun to turn their attention to noradrenaline, a chemical that plays a critical role in brain functions including attention and arousal, thinking and motivation.
Professor James Rowe from the Department of Clinical Neurosciences at the University of Cambridge, who led the study, said, Noradrenaline is very important for brain function. All of our brains supply comes from a tiny region at the back of the brain called the locus coeruleus which means the blue spot. It’s a bit like two short sticks of spaghetti half an inch long: it’s thin, it’s small, and it’s tucked away at the very base of the brain in the brain stem.
-
New research indicates that blocking proteins with medications…
Imaging Studies Can Differentiate Parkinsons From Other Causes Of Parkinsonism
Catherine L. Gallagher, MD
Although Parkinsons disease remains a clinical diagnosis, imaging studies are an important ancillary test for differential diagnosis of movement disorders. Imaging studies may be used to rule out structural and other causes of parkinsonian symptoms. Single-photon emission computed tomography scans using labeled tracers for dopamine transporters can also be used to confirm parkinsonism or differentiate PD from secondary causes of parkinsonian motor symptoms. Finally, imaging studies are being used in research to better understand the pathophysiology of PD and elucidate causative mechanisms that could be therapeutic targets in the future.
Don’t Miss: Symptoms And Progression Of Parkinson’s Disease
Detection Of Preclinical Pd
For every patient who presents with clinical PD there may be 10 subclinical cases with incidental brain stem Lewy body disease in the community . Subjects at risk of developing PD include carriers of genetic mutations known to be associated with parkinsonism , relatives of patients with the disorder, elderly subjects with idiopathic hyposmia, and patients with rapid-eye-movement sleep behavior disorders.
Relatives of PD patients with idiopathic hyposmia are at risk of PD. Ponsen et al. collected 40 such relatives after screening 400 subjects for hyposmia and, with 123I–CIT SPECT, found that 7 of these showed reduced striatal DAT binding . Four of these 7 subsequently converted to clinical PD over a 2-y period.
Patients with idiopathic rapid-eye-movement sleep behavior disorder are at high risk of developing parkinsonism or dementia. Using 123I-IPT SPECT, Eisensehr et al. found reduced striatal DAT binding in all 5 of their patients with idiopathic rapid-eye-movement sleep behavior disorder . In another series, 11 patients with sleep disorders were investigated with FP-CIT SPECT, and reduced striatal DAT binding was found in 3, 1 of whom had evidence of clinical parkinsonism .
Imaging The Pharmacology Of Depression In Pd
The prevalence of depression in PD has been reported to range from 10% to 45% . Because Lewy body pathology is known to affect serotonergic and noradrenergic as well as dopaminergic neurotransmission, dysfunction of any or all of these systems would seem to be a reasonable candidate for the functional substrate of depression . To date, functional imaging has failed to demonstrate a correlation between serotonergic dysfunction and depression in PD.
123I–CIT binds with nanomolar affinity to dopamine, noradrenaline, and serotonin transporters. Although striatal uptake of 123I–CIT 24 h after intravenous injection primarily reflects DAT binding, midbrain uptake 1 h after administration reflects serotonin transporter availability . Kim et al. have reported normal brain stem 123I–CIT uptake in PD. They found no difference between uptake in depressed and nondepressed patients and no correlation between radiotracer uptake and Hamilton Depression Rating Scale scores .
Recommended Reading: Does Parkinson’s Cause Hair Loss
Clinical Application Of Brain Mri In The Diagnostic Work
aDepartment of Radiology and Nuclear Medicine, Radboud University Medical Center, Nijmegen, The NetherlandsbDepartment of Diagnostic Imaging, Medical Center of Postgraduate Education, Warsaw, PolandcDepartment of Neurology, Donders Institute for Brain, Cognition and Behaviour, Radboud University Medical Center, Nijmegen, The Netherlands
Keywords: Atypical parkinsonism, brain, magnetic resonance imaging, Parkinsons disease
ABSTRACT
Background: Differentiating Parkinsons disease and atypical parkinsonism on clinical parameters is challenging, especially in early disease courses. This is due to large overlap in symptoms and because the so called red flags, i.e. symptoms indicating atypical parkinsonism, have not developed. Brain MRI can aid to improve the accuracy and confidence about the diagnosis.Objective and Methods: In the current paper, we discuss when brain MRI should be performed in the diagnostic work-up of parkinsonism, our preferred brain MRI scanning protocol, and the diagnostic value of specific abnormalities.Results and Conclusions: The main purpose of brain MRI is to assess cerebrovascular damage, and to exclude other possible and sometimes treatable causes of parkinsonism, such as normal pressure hydrocephalus. Furthermore, brain MRI can support the possible or probable diagnosis of a specific form of atypical parkinsonism.
INTRODUCTION OF THE CLINICAL DILEMMA
DESCRIPTION OF THE TEST
DISCUSSION
ACKNOWLEDGMENTS
CONFLICT OF INTEREST
Brain Mr Contribution To The Differential Diagnosis Of Parkinsonian Syndromes: An Update
Giovanni Rizzo
1IRCCS Istituto delle Scienze Neurologiche, Bellaria Hospital, Bologna, Italy
2Neurology Unit, Department of Biomedical and Neuromotor Sciences, University of Bologna, Bologna, Italy
3Functional MR Unit, Policlinico S.Orsola-Malpighi, Bologna, Italy
4Department of Biomedical and Neuromotor Sciences, University of Bologna, Bologna, Italy
5Department of Diagnostic Imaging, Pia Fondazione di Culto e Religione Card. G. Panico, Tricase, Italy
6Department of Neurology, Kings College NHS Foundation Trust, London, UK
7Department of Neurology, Queen Elizabeth Hospital, Lewisham and Greenwich NHS Trust, London, UK
8Department of Neurology and Health Science Research, Mayo Clinic, Rochester, MN, USA
9Department of Clinical Research in Neurology, University of Bari, Pia Fondazione di Culto e Religione Card. G. Panico, Tricase, Italy
10Department of Basic Medical Science, Neuroscience and Sense Organs, University of Bari, Bari, Italy
Abstract
1. Introduction
Many putative biomarkers derived from genetic-epigenetic, neurophysiological, and imaging techniques have been evaluated in order to determine their diagnostic accuracy in discriminating PD from APSs . Brain magnetic resonance represents one of the best putative sources of biomarkers in this field, because of its relative feasibility, the absence of invasiveness, and the availability in different clinical settings.
2. Qualitative Brain MRI
3. Quantitative Brain MR
| MSA |
| NAA/mI |
3.1. PD
3.2. MSA
Recommended Reading: Which Part Of The Brain Is Affected By Parkinson’s Disease