Understanding The Neurologic Control Of The Cardiac System
Before we explore this issue, lets first learn a bit about the autonomic nervous system and about the cardiac systems place within it. The ANS is part of the peripheral nervous system, a network of nerves throughout the body. The ANS exerts control over functions that are not under conscious direction such as respiration, heart function, blood pressure, digestion, urination, sexual function, pupillary response, and much more. The ANS is further subdivided into the parasympathetic nervous system and the sympathetic nervous system. Both the parasympathetic and sympathetic nervous systems regulate most major organs. Often, they have opposite effects, with the sympathetic nervous system activating a system and the parasympathetic system calming it down.
One of the systems controlled by the ANS is cardiac regulation. Blood pressure sensors, known as baroreceptors, reside in the heart as well as in the carotid artery, the major artery in the neck. If the baroreceptors sense a change in the blood pressure, a signal is sent to particular areas in the brain. From there, the autonomic nervous system sends signals to the heart to control heart rate and cardiac output. Signals are also sent to the blood vessels to change the size of their diameter, thereby regulating blood pressure.
Treatments For Wpw Syndrome
In many cases, episodes of abnormal heart activity associated with WPW syndrome are harmless, don’t last long, and settle down on their own without treatment.
You may therefore not need any treatment if your symptoms are mild or occur very occasionally, although you should still have regular check-ups so your heart can be monitored.
If your cardiologist recommends treatment, there are a number of options available. You can have treatment to either stop episodes when they occur, or prevent them occurring in the future.
The Heart Of The Matter: Cardiovascular Effects Of Parkinsons Disease
It has long been understood that Parkinsons disease does not just cause movement symptoms, but also causes a litany of non-motor symptoms with effects throughout the body. One of the organ systems that is affected is the cardiac system, encompassing the heart, as well as the major and minor blood vessels. I received this topic as a suggestion from a blog reader and we will be discussing this important issue today. Please feel free to suggest your own blog topic.
Don’t Miss: How Long Does Parkinson’s Take To Develop
Ans Dysfunction In The Aspect Of Association Between Af And Pd
According to numerous neuroimaging studies, autonomic dysfunction is a common non-motor symptom of PD., Cardiac noradrenergic denervation is one of the pathophysiological basis for this phenomenon. Studies have presented that cardiac noradrenergic denervation is a widespread phenomenon in PD regardless of clinical ANS dysfunction., , In addition, atrial conduction time was prolonged in newly diagnosed patients with PD, and it had a positive correlation with PD severity and duration. Although the pathogenesis of AF is very complex and not fully understood, ANS has an important role in triggering and maintaining AF. Both the sympathetic and parasympathetic nervous systems carry out different roles in the regulation of atrial electrophysiological properties, and the concept of autonomic nervous imbalance has been proposed to explain the atrial electrical instability and AF occurrence in the aspect of cardiac ANS. Furthermore, frequent atrial ectopic beats and atrial conduction time prolongation are well-known predictors of paroxysmal AF., Putting these findings together, cardiac sympathetic denervation and ANS dysfunction are plausible mechanisms of the increased risk of AF in patients with PD.
What Are The Signs And Symptoms Of Wolff
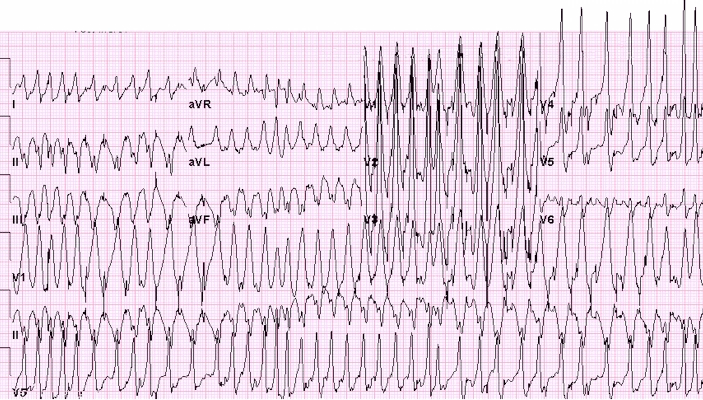
Individuals affected by Wolff-Parkinson-White syndrome can experience palpitations, rapid heart rates, difficulty breathing, and lightheadedness as well as near loss of consciousness and complete loss of consciousness. For the most part, these symptoms occur all of a sudden and are not associated with warning signs. Usually, there are no dramatic triggers, however, caffeine, alcohol, and exercise can cause the heart to start racing.
Also Check: How Does Smoking Lower The Risk Of Parkinson Disease
Role Of The Intrinsic Atrial Muscle Vulnerability
Figure 1Open in new tab
Atrial endocardial mapping sites. The upper part of the figure shows 12 endocardial mapping sites in the right atrium. The atrial endocardial electrograms were recorded in each patient from the anterior, lateral, posterior, and medial aspects of the high right atrium , mid right atrium , and low right atrium . SVC, superior vena cava IVC, inferior vena cava Ao, aorta PA, pulmonary artery LA, left atrium RV, right ventricle LV, left ventricle. The lower part of the figure shows two atrial endocardial electrograms to distinguish an abnormal atrial electrogram with 10 fragmented deflections and 130 ms in duration, from a normal atrial electrogram with two deflections and 80 ms in duration. Reprinted with permission from Centurion et al .
Defining The Comorbidities And Outcome
Comorbidities were defined using ICD-10-CM codes: hypertension , diabetes mellitus , dyslipidemia , congestive heart failure , peripheral artery disease , previous history of myocardial infarction and ischemic stroke , chronic obstructive pulmonary disease , and end-stage renal disease . The primary outcome was the development of non-valvular AF . We regarded the AF with rheumatic mitral stenosis and mechanical heart valves as valvular AF and excluded as in our previous study. We used the same definition of AF reported in previous studies., This operational definition of AF was validated, and the positive predictive value was 94.1% compared with electrocardiogram. Definitions of comorbidities, medications, and outcomes are described in detail in Supplementary Table .
Don’t Miss: Levodopa And The Progression Of Parkinson’s Disease
Pearls And Other Issues
Patients with atrial fibrillation and rapid ventricular response are often treated with amiodarone or procainamide. Procainamide and cardioversion are accepted treatments for conversion of tachycardia associated with Wolff Parkinson White syndrome . In acute AF associated with WPW syndrome, the use of IV amiodarone may potentially lead to ventricular fibrillation in some reports and thus should be avoided.
AV node blockers should be avoided in atrial fibrillation and atrial flutter with Wolff Parkinson White syndrome . In particular, avoid adenosine, diltiazem, verapamil, and other calcium channel blockers and beta-blockers. They can exacerbate the syndrome by blocking the heart’s normal electrical pathway and facilitating antegrade conduction via the accessory pathway.
An acutely presenting wide complex tachycardia should be assumed to be ventricular tachycardia if doubt remains about the etiology.
How Is Wpw Treated
Treatment depends on the type and frequency of arrhythmias, associated symptoms such as syncope, and presence of structural heart disease. Typically a physician will recommend an ablation procedure to further define the characteristics of the accessory pathway, and ultimately, to eliminate the pathway entirely.
- Observation – If you have no symptoms, you may not require treatment. Your doctor may choose to have regular follow-up without treatment.
- Medications – A variety of drugs are available to treat arrhythmias. Because everyone is different, it may take trials of several medications and doses to find the one that works best for you. It is important to know:
- The names of your medications
- What they are for
- How often and at what times to take them
Don’t Miss: Foods Not To Eat With Parkinson’s Disease
Different Anatomical Sites Of The Accessory Pathway
The location of the AP was also related to the induction of AF. It was shown that patients with an anteroseptal AP had a high rate of inducible arrhythmia . Patients with a right free wall AP had a rather low rate of inducible arrhythmia . Patients with left free wall and posteroseptal AP had a 44 and 36% rate of induction, respectively. Patients with a right-sided AP had a lower inducibility of AVRT and a relatively long retrograde ERP over the AP. This allowed only relatively late PVCs to be conducted retrogradely over the AP to the atria, which might explain the lower rate of inducibility of AF in these patients.
Enhancing Healthcare Team Outcomes
AF is a huge financial burden on health care. Therefore, managing AF requires an interprofessional approach with close involvement of various health care professionals such as primary care physicians, cardiologists, electrophysiologists, neurologists, surgeons, pharmacists, and nursing staff.
Ensuring guideline-directed treatment of AF is pivotal in improving overall outcomes and reducing health care costs. Nurse-led, guideline-based clinics supported by appropriate software and cardiologists have shown superior results in cardiovascular mortality and hospitalizations. . Outpatient specialty AF clinics have shown better outcomes in AF-related hospitalizations and quality of life compared to usual care clinics. . Similar results have been seen with pharmacist-managed anticoagulation compared to telephone/usual care clinics. AF management guidelines published by the European Society of Cardiology have stressed the importance of integrated management in coordinating care and improving outcomes.
Also Check: How Do You Get Parkinson’s
How Parkinsons Disease Affects The Autonomic Nervous System And The Heart
In PD, there are two major reasons why the automatic control of the cardiac system is impaired. First, areas of the brain that control this system often contain Lewy bodies and have undergone neurodegeneration. In addition, the autonomic nervous system itself is directly affected by Lewy body-like accumulations and neurodegeneration. This means, when the baroreceptors in the heart and carotid artery sense a drop in blood pressure and try to generate a signal to the heart and blood vessels to increase the blood pressure, the message may not get through. This results in neurogenic orthostatic hypotension , or drops in blood pressure upon standing due to autonomic nervous system dysfunction. There are no medications that can cure nOH by restoring the autonomic nervous system in PD. nOH however, can be treated. Read more about nOH and its treatments here.
Structural problems of the heart such as coronary artery disease or cardiomyopathy are not thought to be part of the pathology of PD, although of course, could co-exist with PD.
Treatment Of Atrial Fibrillation And Wpw Syndrome
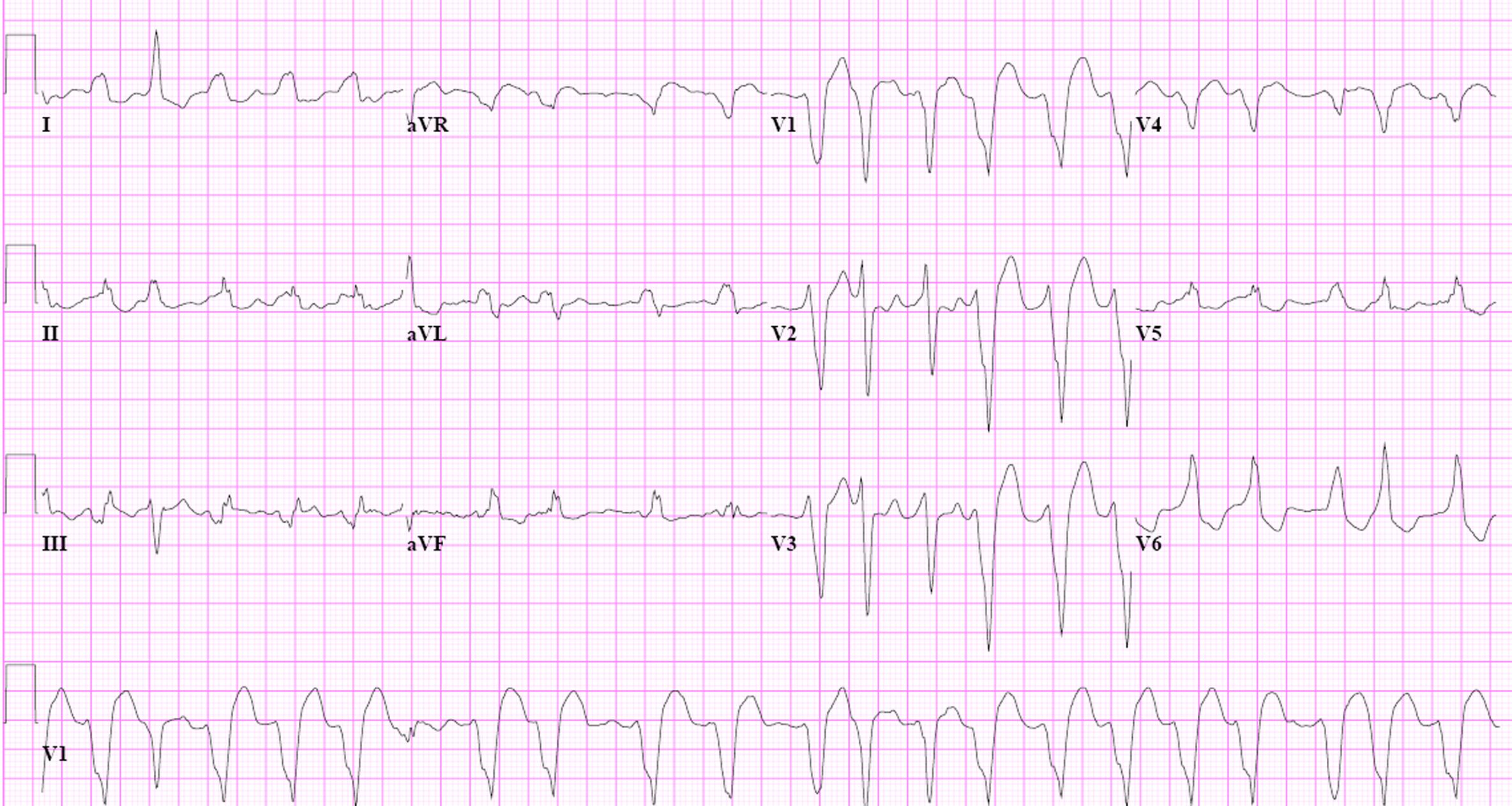
-
Direct-current cardioversion
The treatment of choice for Wolff-Parkinson-White syndrome is direct-current cardioversion Direct-Current Cardioversion-Defibrillation The need for treatment of arrhythmias depends on the symptoms and the seriousness of the arrhythmia. Treatment is directed at causes. If necessary, direct antiarrhythmic therapy, including antiarrhythmic… read more . The usual rate-slowing drugs used in atrial fibrillation are not effective, and digoxin and the nondihydropyridine calcium channel blockers are contraindicated because they may increase the ventricular rate and cause ventricular fibrillation. If cardioversion is impossible, drugs that prolong the refractory period of the accessory connection should be used. IV procainamide or amiodarone is preferred, but any class Ia, class Ic, or class III antiarrhythmic drug Drugs for Arrhythmias The need for treatment of arrhythmias depends on the symptoms and the seriousness of the arrhythmia. Treatment is directed at causes. If necessary, direct antiarrhythmic therapy, including antiarrhythmic… read more can be used.
You May Like: Perioperative Management Of Parkinson Disease
Prevention Of Thromboembolism During Rhythm Control
Patients, particularly those in whom the current episode of atrial fibrillation has been present > 48 hours, have a high risk of thromboembolism for several weeks after pharmacologic or direct current cardioversion. If the onset of the current episode of atrial fibrillation is not clearly within 48 hours, the patient should be anticoagulated for 3 weeks before and at least 4 weeks after cardioversion regardless of the patient’s predicted risk of a thromboembolic event .
Alternatively, therapeutic anticoagulation is started, transesophageal echocardiography is done, and, if no left atrial or left atrial appendage clot is seen, cardioversion may be done, followed by at least 4 weeks of anticoagulation therapy.
If urgent cardioversion is required because of hemodynamic compromise, cardioversion is done and anticoagulation is started as soon as is practical and continued for at least 4 weeks.
If the onset of the current episode of atrial fibrillation is clearly within 48 hours, cardioversion may be done without prior anticoagulation if the patient has nonvalvular atrial fibrillation and is not at high risk of a thromboembolic event. After cardioversion, therapeutic anticoagulation is given for 4 weeks however, anticoagulation may not be necessary in patients at low risk of a thromboembolic event.
After 4 weeks of postconversion anticoagulation therapy, some patients require long-term anticoagulation .
Baseline Characteristics Of The Cohort
Study participants were followed up until December 31, 2017, and the mean follow-up duration was 3.35 ± 1.8 years. The baseline characteristics of the study population are summarized in Table . The mean age of all the participants was approximately 70 years and 40.2% of them were men. The proportion of patients with low income was higher in the PD group than in the control group . Compared with patients without PD, the patients with PD had greater number of comorbidities, including hypertension, DM, dyslipidemia, CHF, previous MI, stroke, PAD, COPD, and ESRD.
| Parkinsonâs disease |
|---|
You May Like: Motor And Non Motor Symptoms Of Parkinson’s Disease
What Causes Wpw Syndrome
When the heart beats, its muscular walls contract to force blood out and around the body. They then relax, allowing the heart to fill with blood again. This is controlled by electrical signals.
In WPW syndrome, there’s an extra electrical connection in the heart, which allows electrical signals to bypass the usual route and form a short circuit. This means the signals travel round and round in a loop, causing episodes where the heart beats very fast.
The extra electrical connection is caused by a strand of heart muscle that grows while the unborn baby is developing in the womb.
It’s not clear exactly why this happens. It just seems to occur randomly in some babies, although rare cases have been found to run in families.
Propensity Score Matching Results
After propensity score matching, the baseline characteristics between the PD and control groups became equivalent . The distribution of the propensity score after matching was well-balanced and assessed by standardized differences of baseline characteristics . In line with our main results, the risk of AF was significantly higher in the PD group than in the control group . Subgroup analyses were performed using the propensity score-matched population . Significant interaction was consistently observed in the subgroup of patients with hypertension after propensity score matching. The subgroup with diabetes showed significant interaction after matching. However, subgroups with previous stroke and one or more cardiovascular risk factors did not show significant interaction after propensity score matching.
Recommended Reading: Michael J Fox Parkinson’s Website
Abnormal Atrial Endocardial Electrograms In Sinus Rhythm
Figure 5Open in new tab
Normal atrial endocardial electrograms. Examples of measurement of the duration , and the number of deflections of the intraatrial electrogram. Surface electrocardiographic lead V1 is shown together with the right atrial electrogram . In the top panel, the arrows show the onset and the end of the intraatrial electrogram. In the bottom panel, the arrows show the deflections of the intraatrial electrograms. Reprinted with permission from Konoe A, Fukatani M, Tanigawa M, et al . Electrophysiological abnormalities of the atrial muscle in patients with manifest WolffParkinsonWhite syndrome associated with paroxysmal atrial fibrillation. PACE 1992 15:10401052. Abnormally prolonged and fractionated atrial endocardial electrogram. Example of an abnormal atrial electrogram. This abnormal atrial electrogram was recorded from high lateral right atrium . Abnormal atrial electrogram as defined in the text. Reprinted with permission from Konoe et al .
Warning Disclaimer Use For Publication
WARNING: Please DO NOT STOP MEDICATIONS without first consulting a physician since doing so could be hazardous to your health.
DISCLAIMER: All material available on eHealthMe.com is for informational purposes only, and is not a substitute for medical advice, diagnosis, or treatment provided by a qualified healthcare provider. All information is observation-only. Our phase IV clinical studies alone cannot establish cause-effect relationship. Different individuals may respond to medication in different ways. Every effort has been made to ensure that all information is accurate, up-to-date, and complete, but no guarantee is made to that effect. The use of the eHealthMe site and its content is at your own risk.
If you use this eHealthMe study on publication, please acknowledge it with a citation: study title, URL, accessed date.
Read Also: Parkinson’s Dbs Side Effects
Atrial Electrophysiological Responses Induced By Programmed Stimulation
AF can also be initiated by ectopic beats originating from the pulmonary veins and elsewhere. The pulmonary veins are well established as the dominant sources of triggers in PAF, in addition to their contribution to maintenance of AF. However, there is no available data suggesting that firing from the pulmonary veins is the main source of recurrent AF in WPW patients that had their AP ablated. The elimination of triggers of AF requires spontaneous firing to be readily identifiable during an ablation procedure. Ablation targeting the pulmonary veinleft atrial junction is effective in isolating the left atrium from proarrhythmic pulmonary vein activity. Despite the latest progress in AF ablation, there is limited knowledge of how to identify, map, and ablate the culprit atrial substrate in an individual patient, because AF is generally associated with locally complex electrograms of indefinable timing and sequence. This heterogeneity of substrate may explain why no single predetermined ablation technique is effective for all patients across the entire spectrum of AF. To the best of our knowledge, there is no detailed study addressing ablation of the pulmonary veins to suppress recurrent AF in WPW patients that had already undergone successful AP ablation.
How Is Wpw Diagnosed

WPW can only be diagnosed by reviewing an ECG . A holter or ambulatory monitor and exercise testing are also helpful in evaluating patients known to have WPW.
In the past, patients with WPW but without symptoms had been observed by a cardiologist for many years. Recently, new guidelines have been published for this group of patients. Your cardiologist may order a holter monitor or stress test to look for a persistent patter of WPW. If the WPW pattern persists, invasive electrophysiology testing is now recommended.
Your doctor will also ask you several questions:
- Do you have symptoms?
- Do you have a history of atrial fibrillation?
- Do you have a history of fainting?
- Do you have a history of sudden cardiac death or does anyone in your family?
- Are you a competitive athlete?
The results of your diagnostic tests and the answers to these questions will help guide your therapy.
Don’t Miss: Signs Of Parkinson’s Disease Dementia