Symptomatic Pharmacotherapy Of Parkinsonism
Anticholinergics
Anticholinergics were the first drugs for the treatment of PD.
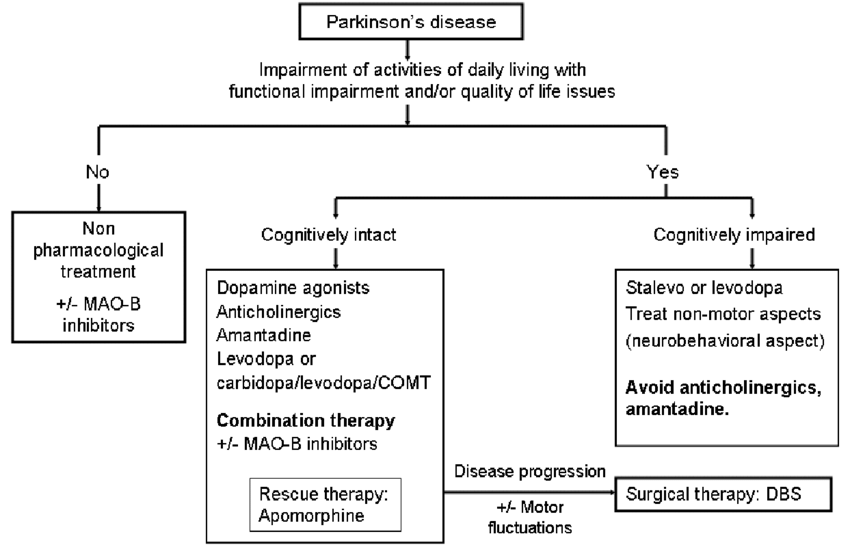
| Patients not on dopaminergic therapy |
| If a patient has started on an MAO-B inhibitor, anticholinergic, amantadine or a combination of these, a stage will come when there is a requirement for adding levodopa or a dopamine agonist |
| Patients on dopaminergic therapy |
| If on dopamine agonist therapy: |
| Increase the dose |
| Switch between agonists |
| Add levodopa |
| Add a COMT inhibitor |
| Patients with disabling tremor |
| Deep brain stimulation |
Monotherapy
Three Class II trials found anticholinergic monotherapy more effective than placebo in improving motor function . Biperiden was as effective as apomorphine for parkinsonian tremor . Two systematic reviews concluded that anticholinergics have a small motor effect, but that evidence for a specific effect on tremor is inconclusive , .
Adjunctive therapy
Class II studies of trihexyphenidyl, benzotropine and bornaprine in levodopa-treated patients and two systematic reviews conclude that there is a minor effect on PD symptoms and that tremor-specific data are inconclusive , .
Prevention of motor complications
No studies available.
Safety
The clinical use of anticholinergics is limited by cognitive and neuropsychiatric side effects .
Amantadine
Amantadine blocks NMDA glutamate receptors and may have an anticholinergic effect and release presynaptic dopamine stores.
Monotherapy
Adjunctive therapy
Prevention of motor complications
No studies available.
Safety
Levodopa
Safety
Medication Guidelines For Parkinson’s Disease
There is no one best mix of Parkinsonâs medicines. You and your doctor will have to try a few treatment approaches to figure out the best one for you.
But there are some general guidelines for taking your medication. Be sure to ask your doctor or pharmacist for any specific tips for your treatment.
Diagnosis Of Parkinson’s Disease
The diagnosis of PD is clinical and requires bradykinesia, defined as slowness of movement and decrement in amplitude or speed, usually assessed using finger tapping, foot tapping or pronationsupination hand movements. In addition, rest tremor or rigidity is required to confirm a parkinsonian syndrome. Tremor was absent at presentation in 30% in one series of pathologically proven PD. Patients with suspected PD should be referred quickly and untreated to a specialist in movement disorders for evaluation. Key points for discussion at diagnosis include the need to inform vehicle licensing agencies and insurers, signposting to written or web-based information on newly diagnosed PD, and provision of contact details for the local PD nurse specialist .
Current International Parkinson and Movement Disorder Society diagnostic criteria for Parkinson’s disease adapted from Postuma RB, Berg D, Stern M et al. MDS clinical diagnostic criteria for Parkinson’s disease. Mov Disord 2015 30:1591601. At least two supportive criteria and no red flags required for a diagnosis of clinically established Parkinson’s disease. Conditions in italics should be considered if the corresponding exclusion criteria or red flags are present.
Recommended Reading: How To Prevent Getting Parkinson’s Disease
Pathophysiology And Presenting Features
Classic presenting features of PD include motor symptoms, such as bradykinesia, rigidity, rest tremor and postural instability. However, non-motor symptoms, such as depression, cognitive impairment, pain and autonomic disturbances, are also often present and they can severely affect a patients quality of life. There are several information sheets available for patients that cover the management of multiple common types of pain in PD.
The motor symptoms are largely caused by the progressive loss of dopaminergic neurons in the substantia nigra compacta, which ultimately reduces dopaminergic input to the striatum and other brain regions. Compensatory mechanisms in the brain are so effective that the clinical symptoms of PD may only develop when around 80% of dopaminergic neurons have degenerated. By contrast, the Braak theory of PD suggests that the disease process starts in the olfactory bulb and lower part of the medulla, and it is not until stage 3 that the substantia nigra becomes involved in the process. There is also direct evidence of Parkinson pathology being spread from the gastrointestinal tract to the brain in rodents. There are therapeutic implications of gut involvement it is known that swallowing and the stomach are the two main problems of PD therapy and lead to the use of non-oral therapies.
Neuroprotection And Disease Modification
MAO-B inhibitors
Selegiline in early PD was shown to postpone the need for dopaminergic treatment by several months. Subjects whose treatment with rasagiline was delayed by 6 months showed greater worsening on UPDRS III .
- a Subcutaneous apomorphine is not used in early PD.
- b Ergot derivates cannot be recommended as a first-line treatment because of the risk of valvular heart disorder.
- c No recommendation can be made because of insufficient data.
- d Transdermal patch.
- e Controlled release.
- fEntacapone and tolcapone should always be given with levodopa. Because of hepatic toxicity, tolcapone is not recommended in early PD.
| The choice of drug depends on the impact of improving motor disability compared with the risk of motor complications and neuropsychiatric complications |
| Options include the following: |
| MAO-B inhibitor |
| Oral or transdermal dopamine agonist. Pramipexole, piribedil, ropinirole and rotigotine are effective . Initial treatment with an agonist can be recommended in younger patients . Ergot derivatives are not recommended as first-line medication because of the risk of fibrotic reactions |
| Levodopa is the most effective symptomatic drug . Controlled-release formulations or adding entacapone is not effective in the delay of motor complications |
| Amantadine or an anticholinergic |
| Rehabilitation: because of the lack of evidence in early-stage disease, a recommendation cannot be made |
Levodopa
Dopamine agonists
There is no robust evidence of disease modification .
Also Check: Parkinson’s Disease Causes Death
A Review On Parkinsons Disease Treatment
5214526Tori K. Lee Eva L. Yankee
Department of Biology, Angwin, CA 94508, USA .
Received:First Decision:Revised:Accepted:Available online:Academic Editors:Copy Editor:Production Editor:
© The Author 2021. Open Access This article is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Motor Fluctuations And Dyskinesia
For the treatment of motor features of tremor, bradykinesia, and rigidity associated with Parkinson’s disease, dopaminergic therapies are initially effective however, motor fluctuations eventually complicate therapy and can cause significant disability and impair quality of life. Sustained-release carbidopa/levodopa and bromocriptine have not been found to reduce off time.
Risk factors for motor complications include disease severity, younger age at onset of Parkinson’s disease, high levodopa dosage, and longer disease duration. The motor fluctuations usually are addressed with levodopa adjustments as well as adjunctive medications or surgery as discussed below.
Read Also: What Tests Are Done To Diagnose Parkinson’s
Parkinsons Disease: Management And Guidance
An overview of Parkinsons disease management, including discussion of the updated National Institute for Health and Care Excellence guidelines.
Nervous system diseases
DR. MICHAEL SOUSSAN/ISM/SCIENCE PHOTO LIBRARY
Parkinsons disease is a chronic, progressive neurodegenerative condition resulting from the loss of the dopamine-containing cells of the substantia nigra, and its prevalence increases with age. Using primary care data from 2015, a Parkinsons UK report of the Clinical Practice Research Datalink found that the prevalence of PD is 45 per 100,000 people who are aged 3039 years, compared with 1,696 per 100,000 people who are aged 8084 years. Prevalence rates almost double at each five-year interval between the ages of 50 and 69 years for both men and women. The lifetime risk of being diagnosed with PD is 2.7% equating to 1 in every 37 people being diagnosed at some point in their lifetime. Owing to population growth and an increasing ageing population, the estimated prevalence of PD is expected to increase by 23.2% by 2025.
Common Drugs For Parkinson’s Disease
Levodopa and carbidopa . Levodopa is the most commonly prescribed medicine for Parkinsonâs. Itâs also the best at controlling the symptoms of the condition, particularly slow movements and stiff, rigid body parts.
Levodopa works when your brain cells change it into dopamine. Thatâs a chemical the brain uses to send signals that help you move your body. People with Parkinsonâs donât have enough dopamine in their brains to control their movements.
Sinemet is a mix of levodopa and another drug called carbidopa. Carbidopa makes the levodopa work better, so you can take less of it. That prevents many common side effects of levodopa, such as nausea, vomiting, and irregular heart rhythms.
Sinemet has the fewest short-term side effects, compared with other Parkinsonâs medications. But it does raise your odds for some long-term problems, such as involuntary movements. An inhalable powder form of levodopa and the tablet istradefylline have been approved for those experiencing OFF periods, OFF periods can happen when Parkinsonâs symptoms return during periods between scheduled doses of levodopa/carbidopa.
People who take levodopa for 3-5 years may eventually have restlessness, confusion, or unusual movements within a few hours of taking the medicine. Changes in the amount or timing of your dose will usually prevent these side effects.
Dopamine agonists. These drugs act like dopamine in the brain. They include pramipexole , rotigotine , and ropinirole , .
Don’t Miss: Why Does Parkinson’s Cause Hallucinations
Manual Therapy And Exercise
Chiropractic manipulation, osteopathic manipulation, and Trager therapy have been suggested to benefit patients with Parkinson’s disease. No studies exist, however, to refute or confirm this position. The Alexander technique has shown some benefit and patient improvement has been noted in some studies.
Standard physical therapy, as well as occupational therapy, did result in improved functional outcomes, but the benefit was small and was not sustained when the exercise therapy stopped.
When To Start Treatment
Deciding when to start drug therapy for Parkinsons disease should be individually tailored to a patients symptoms, circumstances and comorbidities. Treatment is indicated when symptoms impact on quality of life. When treatment is needed there is no evidence to support undue delay because of concerns about levodopa toxicity or the development of treatment resistance.3 The aim is to control symptoms and maintain an on state.
Some drugs with good symptomatic benefit are speculated to have a role in neuroprotection and some specialists advocate their use from the time of diagnosis.4 Delayed start trials have been used to try and differentiate symptomatic from disease-modifying effects. A recent delayed start study of rasagiline, a monoamine oxidase B inhibitor, in treatment-naïve patients with mild Parkinsons disease showed a small benefit in the low-dose treatment group. This was not seen with the 2 mg dose and a clear explanation for this has not been established.5 Further studies are needed before such treatments are considered truly disease modifying. Until a drug is unequivocally proven to slow disease progression, the time to commence treatment will remain contentious.
Don’t Miss: On Off Phenomenon In Parkinson’s Disease
Anticholinergics For Early On
The first pharmacological agents used in PD therapy were anticholinergic drugs. They reduce the activity of acetylcholine by acting as antagonists at choline receptors, hoping to restore the balance between dopamine and acetylcholine levels that was disturbed by PD. These drugs have largely been replaced by L-DOPA and other centrally acting dopaminergic agonists, but they still remain available for use in the treatment of PD. Benztropine, biperiden, diphenhydramine, ethopropazine, orphenadrine, procyclidine, and trihexyphenidyl are included in this therapeutic class of drugs, though there is little pharmacokinetic information available on them because of their low plasma drug concentrations. Typically, anticholinergic drugs have a greater role in tremor-predominant PD and can be a monotherapy in early stages, but are usually done in adjunct with L-DOPA or other prescribed medications.
Pharmacological Treatment Of Parkinson’s Disease
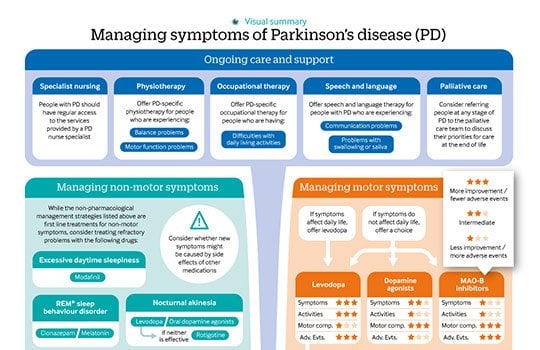
There is currently no proven disease-modifying or neuroprotective therapy for PD. A summary of previous neuroprotection trials is given in a recent review article. Current evidence-based treatment for PD is symptomatic and mainly based around dopaminergic replacement or modulation . The evidence base is summarised in recent guidelines from the National Institute for Health and Care Excellence and the International Parkinson and Movement Disorder Society. Levodopa, dopamine agonists and monoamine oxidase B inhibitors are all licensed for use as initial therapy in PD. Anticholinergics are no longer routinely used due to the risk of cognitive decompensation.
Pharmacological therapies currently used for initial and adjunctive treatment of motor symptoms in Parkinson’s disease
Also Check: Parkinson’s Age Of Onset
Basics Of Parkinsons Disease
Parkinsons disease , or paralysis agitans, is a common neurodegenerative condition, which typically develops between the ages of 55 and 65 years. This disease was first named and described by James Parkinson in 1817. The progression of this disease is gradual and prolonged. It has a plausible familial incidence, although the estimates of these occurrences are low and usually sporadic. This disease is organized into two classifications: genetic and sporadic. Genetic PD follows Mendelian inheritance. Sporadic PD, which accounts for about 90% of all Parkinsons cases, is a more complex category in which the pathogenic mechanisms that underlie it are not yet fully understood. Nonetheless, it is known that the byzantine interactions of genetic and environmental influences play roles in the determination of sporadic PD. Several subtypes of PD exist. Each has its own set of causative factors and susceptibilities, pathology, and treatment courses. General risk factors, symptoms, and pathology will be discussed first, before addressing some of the subtypes.
Systematic Review: Efficacy And Safety Of Medical Marijuana In Selected Neurologic Disorders
Current systematic review. Endorsed by the American Autonomic Society, the American Epilepsy Society, the Consortium of Multiple Sclerosis Centers, the International Organization of Multiple Sclerosis Nurses, and the International Rett Syndrome Foundation. Reaffirmed January 21, 2014, and January 11, 2020.
Also Check: Latest Cure For Parkinson’s Disease
Canadian Guidelines On Pd
The Canadian Guidelines on PD were developed to address the need to provide health care providers who treat individuals with PD with a tool to guide the best management of the disease. The CGPD are the result of a joint effort by specialists in the treatment of movement disorders, family physicians, allied health professionals, individuals diagnosed with PD and patient advocacy groups such as Parkinson Society Canada. The aim of the CGPD is to improve the care for all Canadians who have been diagnosed with PD through the application of
-
best published evidence,
-
patient involvement in the choice of treatment and informed decision-making, and
-
relevance to the Canadian health care system.
A comprehensive description of the guideline development process is provided with the publication of the CGPD in the Canadian Journal of Neurological Sciences. Since there are other current, high-quality guidelines available for the management of PD, the goal of the CGPD was to review these guidelines to select the recommendations most relevant to the Canadian health care provider.
Treating Parkinsons With Complementary Medicine
Complementary medicine incorporates many different practices that can be used alongside conventional medicine to try to ease PD symptoms. There is typically not as much rigorous data to support the use of complementary medicine techniques, as compared to conventional medicine, but many patients find them helpful. These include yoga and massage.
Read Also: Iowa Parkinson’s Disease Association
Aan Releases Recommendations On Treatment Of Parkinson’s Disease
Am Fam Physician. 2007 Mar 15 75:922-924.
-
Guideline source: American Academy of Neurology
-
Literature search described? Yes
-
Evidence rating system used? Yes
-
Available at:
Parkinson’s disease is the second most common neurodegenerative disease and is characterized by bradykinesia tremor at rest rigidity and abnormalities of balance, posture, and gait. Its etiology remains unknown in most patients. Recommendations from the Quality Standards Subcommittee of the American Academy of Neurology discuss the following aspects of this condition in a collection of articles in the April 2006 issue of Neurology: diagnosis and prognosis neuroprotective strategies and alternative therapies treatment and evaluation and treatment of depression, psychosis, and dementia in patients with Parkinson’s disease.
Advanced Therapies For Parkinson’s Disease
When significant off-time or dyskinesia persists despite optimised oral treatment, advanced therapies should be considered. Apomorphine, a potent dopamine agonist given by continuous subcutaneous infusion, is the least invasive and most straightforward of these. Following on from extensive clinical experience, the recent double-blind TOLEDO study confirmed a significant reduction in off-time and improved on-time with apomorphine versus placebo.
Deep-brain stimulation involves surgical placement of electrodes into brain regions such as the subthalamic nucleus to improve motor fluctuations or refractory tremor. DBS is typically considered in patients without significant axial or neuropsychiatric problems. It improves motor function, off-time and QoL in patients with PD, and provides significant benefits over medical therapy even in patients with an average disease duration of 7 years. The benefits of DBS on motor function, fluctuations and activities of daily living have been demonstrated up to 10 years postoperatively, although axial features continue to progress. Infusion of levodopacarbidopa intestinal gel via jejunostomy has also been shown to improve off-time compared to oral levodopa, and is commissioned in specialist centres where other advanced therapies are ineffective or contraindicated. Further information on the decision-making process for advanced therapies is summarised in a dedicated review.
Don’t Miss: Parkinson’s Disease And Driving
Editorial Note On The Review Process
F1000 Faculty Reviews are commissioned from members of the prestigiousF1000 Faculty and are edited as a service to readers. In order to make these reviews as comprehensive and accessible as possible, the referees provide input before publication and only the final, revised version is published. The referees who approved the final version are listed with their names and affiliations but without their reports on earlier versions .
The referees who approved this article are:
-
Fredric P. Manfredsson, Parkinson’s Disease Research Unit, Department of Neurobiology, Barrow Neurological Institute, Phoenix, Arizona, USA
No competing interests were disclosed.
-
Tipu Z. Aziz, Nuffield Department of Clinical Neurosciences, University of Oxford, Oxford, UK
No competing interests were disclosed.