Clinical Trials For Stem Cell
Table 3. Summary of clinical studies in cell transplantation for PD.
Figure 1. Systematic analysis of various factors associated with clinical outcomes using positron emission tomography readings of Parkinsons disease patients with fetal ventral mesencephalic cell transplantation. Statistical comparison was performed on various parameters against fold change of PET readings pre- and post-transplantation. Age on onset: old vs. young PD patients. Disease stage in mild and severe conditions. Disease duration: long vs. short . Student t-test, *p< 0.05, ***p< 0.001.
Figure 2. Systematic analysis of various factors associated with clinical outcome in fVM cell transplantation in PD patients using UPDRS motor scores. Statistical comparison was performed on varying parameters against the Unified Parkinson Disease Rating Scale motor scores of pre- and post-transplantation. Age on onset: old vs. young PD patients. Disease stage in severe vs. mild condition. Disease duration: long vs. short . Two-way ANOVA, Sidaks multiple comparisons test, **p< 0.005, ***p< 0.001.
Neural Transplantation For The Treatment Of Huntingtons Disease
European Neurological Review,
Abstract:
Overview
Neural transplantation studies where foetal striatal tissue is grafted into the striatum of patients with Huntingtons disease have taken place at several sites worldwide in recent years, following success in rodent models of the disease. Studies have for the most part been safe but have had various degrees of effectiveness. This article looks at the successes and failures of these studies and considers what has been learnt in terms of safety, techniques and methodology. While knowledge of the optimal protocol is advancing, there are still many aspects that need refining, such as immunosuppression and grafting technique. Although advances in this field are hampered by the need for more complete knowledge of the disease itself, the future of neural transplantation has a great deal of potential.Acknowledgement: The authors of this review were involved in the The European Network for Striatal Transplantation in Huntingtons Disease study by Rosser et al. , with patients receiving long-term follow-up at the Cambridge Centre for Brain Repair from Roger Barker. The authors work has been supported by the Medical Research Council and a Biomedical Research Centre award to the University of Cambridge and Addenbrookes Hospital.
Keywords
Huntingtons disease, neural transplantation, striatum
Article:
Results of Huntingtons Disease Trials
Kopyov et al., Los Angeles5
Hauser et al., Florida14
Stem Cell Treatments In The Future Of Parkinsons Disease Management
Although there are a number of challenges brought about with stem cell-based treatments for PD, it seems probable that these treatments will progress to the clinic in the short- to medium-term future. While development of optimized products has been necessarily slow and iterative, the field is now asking questions about how these treatments can be scaled and deliveredthis demonstrates the progress that has been made with these approaches.
As has been discussed, the purpose of stem cell treatments is predominantly to treat the motor symptoms of PD. They will not have any disease-modifying effect and will not treat the major non-motor symptoms which can be particularly disabling in some patients. While these techniques can form one arm of the future of PD treatment, they will likely be combined with other novel treatments targeting alpha-synuclein pathology . It may be possible for stem cell-based regenerative therapies to be employed to restore the function of dopaminergic neurons that have already been lost, while novel disease-modifying drugs could be used to prevent ongoing neuronal death.
You May Like: Best Treatment For Parkinson’s
Assessment Of The Efficacy Of Cell Transplants With Imaging
Last, concurrent with the high demand for the optimization of cell graft visualization in PD, growing emphasis has been placed on enhancing the sensitivity and precision of the spatiotemporal resolution of functional neuroimaging. En route to successful cell transplantation as a therapeutic regenerative method for Parkinsons disease, neuroimaging techniques have to be employed for better patient care. Some key features required to elucidate the therapeutic efficacy of transplanted cells for clinical diagnostics are innervation, survival, differentiation, and functional biochemistry composition. Furthermore, it is crucial that these imaging techniques are time efficient, safe, non-invasive, and allow repeated measures in an individual to determine longitudinal post-operative progression in patients with cell transplantation . In this section, we summarize the pros and cons of current imaging modalities used in tracking cell grafts in PD and their respective biomarkers .
Table 2. Imaging modalities used in cell transplantation for PD.
Induced Pluripotent Stem Cells And Parkinsons Disease
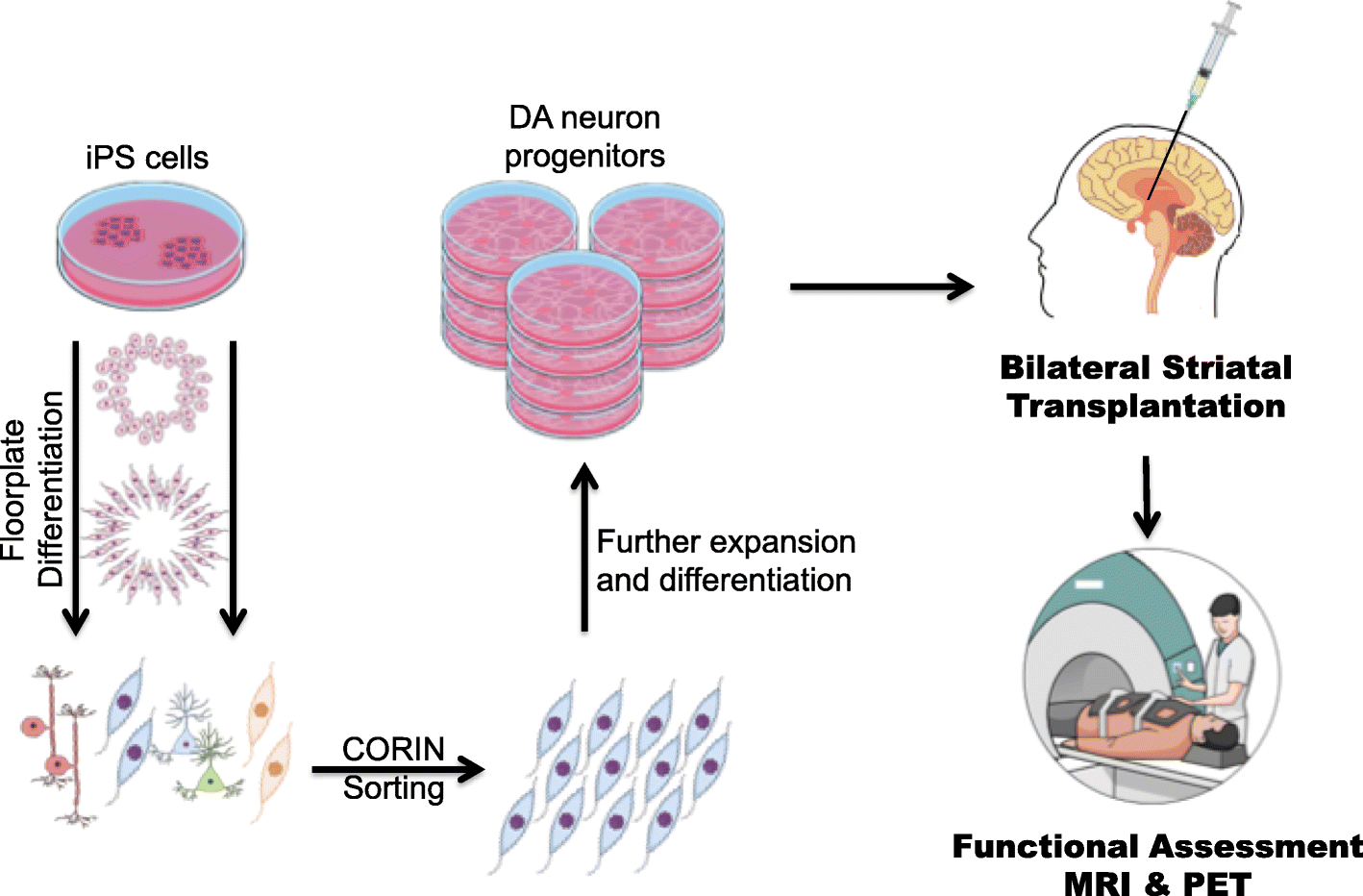
In the early cell therapy, the treatment of central nervous system diseases is to induce neural stem cells to differentiate into specific nerve cells, and then transplant them into the damaged area of the brain, which can improve the dysfunction caused by nerve damage in this area to a certain extent.
For cell transplantation therapy to enter the clinical application of PD, a ready-made cell source with self-renewal potential is essential, and it needs to have the potential to differentiate into DN after transplantation. If DN can be produced from non-dopaminergic cell sources through stem cell differentiation in vitro, and then transplanted into the brain to regulate the release of DA in the striatum, it is expected to limit the disease process of PD patients and help them restore motor function.
Don’t Miss: What Causes Death In Parkinson’s
Speak With Your Doctor About What Parkinsons Treatment Is Right For You
Many things will dictate the best Parkinsons treatment for you, including your symptoms and how advanced it is. Talk to your doctor about possible courses of action, such as medication or a surgical or nonsurgical procedure.
If you found this article informative, be sure to check out some of our others before you go.
Fetal Neural Transplantation As A Treatment For Parkinson’s Disease
Jocelyn Lee
Writers comment: As a student majoring in biochemistry, I never thought neuroscience would inspire me so greatly. A video shown in my NPB class revealed the devastating effects of Parkinsons Disease and motivated me to do further research. After talking to my professor, Charles Gray, and reading several papers and books, I realized that there are promising surgical treatmentsone of them fetal neural transplantationwith the potential to relieve PD symptoms and improve the quality of patients daily lives. Though it may take years or decades before this procedure becomes common clinical practice, it gives millions of Americans with PD a good reason to hope. I am grateful to Dr. Gray for his inspiring lectures and guidance to my English instructor, Anne Fleischmann, for her willingness to help me become a more confident writer and to Palma Lower for encouraging me to submit this paper to Prized Writing.– Jocelyn Lee
Background
Results from Clinical Trials
Long-term Symptomatic Relief
Surgical Criteria/Optimal Fetal Age
Animal experiments have shown that donor age is of crucial importance for the survival of grafted fetal tissue. Neural tissues have to be immature in order to survive transplantation because of their better resistance than adult nervous tissue to the lack of oxygen. The optimal human fetal age is from 6.5 to 9 weeks postconception fetal tissue older than 12 weeks does not survive transplantation in significant quantities .
Patient Selection
Recommended Reading: Does David Brooks Have Parkinson’s
Transplantation For Parkinsons Disease Dont Count It Out Yet
Posted:
Dr. Harold Robertson
Transplantation for treatment of Parkinsons disease has not been as widely discussed as it once was, but dont count it out yet! During the past decade there has been increasing evidence that early diagnosis and treatment of Parkinsons disease will be important in the future. One area where early diagnosis will certainly make a difference is neural transplantation.
A recent issue of Nature drew attention to an article from Ole Isacsons lab at Harvard. The article appeared in the high-impact Journal Cell Stem Cell and is entitled Successful Function of Autologous iPSC -Derived Dopamine Neurons following Transplantation in a Non-Human Primate Model of Parkinsons Disease. This represents a significant step towards a potential treatment or even a cure for Parkinsons disease . In the Maritimes, we have heard very little about transplantation of dopamine neurons since the departure of Dr. Ivar Mendez, so it is worth reviewing the current situation.
Hallett, P.J., Deleidi, M., Astradsson, A., Smith, G.A., Cooper, O., Osborn, T.M., Sundberg, M., Moore, M.A., Perez-Torres, E., Brownell, A.L., et al. . Successful Function of Autologous iPSC-Derived Dopamine Neurons following Transplantation in a Non-Human Primate Model of Parkinsons Disease. Cell Stem Cell.
Barker, R.A., Barrett, J., Mason, S.L., and Bjorklund, A. . Fetal dopaminergic transplantation trials and the future of neural grafting in Parkinsons disease. The Lancet Neurology 12, 84-91.
Induced Pluripotent Stem Cells
In 2007, the process for generating iPSCs was first reported, offering a new avenue for the development of a stem cell-based treatment for PD . iPSCs are generated by the reprogramming of an adult somatic cell into a stem cell, through the expression of a number of transcription factors that could induce pluripotency . The iPSCs derived in this way can be differentiated into dopaminergic neurons using protocols similar to those used with ESCs, which could serve as the basis of a useful cell-based treatment for PD . The potential advantage of iPSC-derived over ESC-derived grafts is that it would be possible to generate autologous grafts, by using a patients own fibroblasts to produce a neural grafting product, negating the requirement for immunosuppression that will be necessary with ESC-derived grafts. However, there are other biological and logistical challenges faced with the iPSC approach, which are discussed below.
iPSC-derived neural grafts have been trialed in primates with MPTP-induced nigral toxicity, with promising results . The neural progenitors grafted ultimately extended neurites into the striatum, did not form any tumors, and resulted in improved motor function at two years. As with the ESC-approach, clinical trials in humans are on the horizon and will begin in the next couple of years .
Recommended Reading: Is There A Parkinson’s Gene
Cell Assessment Of Differentiated Da Neurons
Understanding the key type of DA neurons required to achieve downstream restoration of PD pathology is essential. The mesotelencephalic DA system in the midbrain contains two main groups: the A9 neuronal clusters of the nigrostriatal DA pathway located in the zona compacta, the substantia nigra involved in the control of posture, and the A10 neurons located in the ventromedial mesencephalic tegmentum that regulates the locomotor activity and emotional behavior . Dysfunction of the nigrostriatal system has been linked to Parkinsonism and later to schizophrenia, drug addiction, and depression . Differences between the two DA cell populations have been observed in neurochemistry and in spontaneous neuronal firing . More importantly, A9 neurons display significantly enhanced levels of neuromelanin pigmentation as compared to other dopamine-producing neurons . This could account for the association of early loss of A9 DA neurons in Parkinsons disease with increased vulnerability upon disease progression with the relative preservation of A10 DA neurons .
Generally, stem cells are differentiated into specific nigra A9 DA neurons in large quantities prior to PD transplantation. This step has been thoroughly reviewed by many articles such as in Fan et al. and, thus, will not be further discussed here. However, we focus on developments in technology in cell assessment of differentiated DA neurons.
Evaluation Of Patients For Stereotactic Surgery
Good surgical outcomes from stereotactic surgery for Parkinson disease begin with careful patient selection and end with attentive, detail-oriented postoperative care. The authors believe that this level of care is best provided by a multidisciplinary team comprising a movement disorder neurologist, a neurosurgeon who is well-versed in stereotactic technique, a neurophysiologist, a psychiatrist, and a neuropsychologist. Additional support from neuroradiology and rehabilitation medicine is essential.
At the authors movement disorder center, patients are evaluated for surgery according to the following steps:
-
Evaluation by a neurologist
Michele Tagliati, MD Director of Movement Disorders Program, Cedars-Sinai Medical CenterMichele Tagliati, MD is a member of the following medical societies: American Academy of Neurology, American Medical Association, International Parkinson and Movement Disorder SocietyDisclosure: Nothing to disclose.
Ron L Alterman, MD Associate Professor of Neurosurgery, Mount Sinai School of Medicine Consulting Surgeon, Department of Neurosurgery, Mount Sinai School of Medicine, Elmhurst Hospital, and Walter Reed Army Medical CenterRon L Alterman, MD is a member of the following medical societies: Alpha Omega Alpha, American Association of Neurological Surgeons, Medical Society of the State of New York, New York County Medical Society, Congress of Neurological SurgeonsDisclosure: Nothing to disclose.
Disclosure: Nothing to disclose.
Don’t Miss: Does Parkinson’s Make You Tired
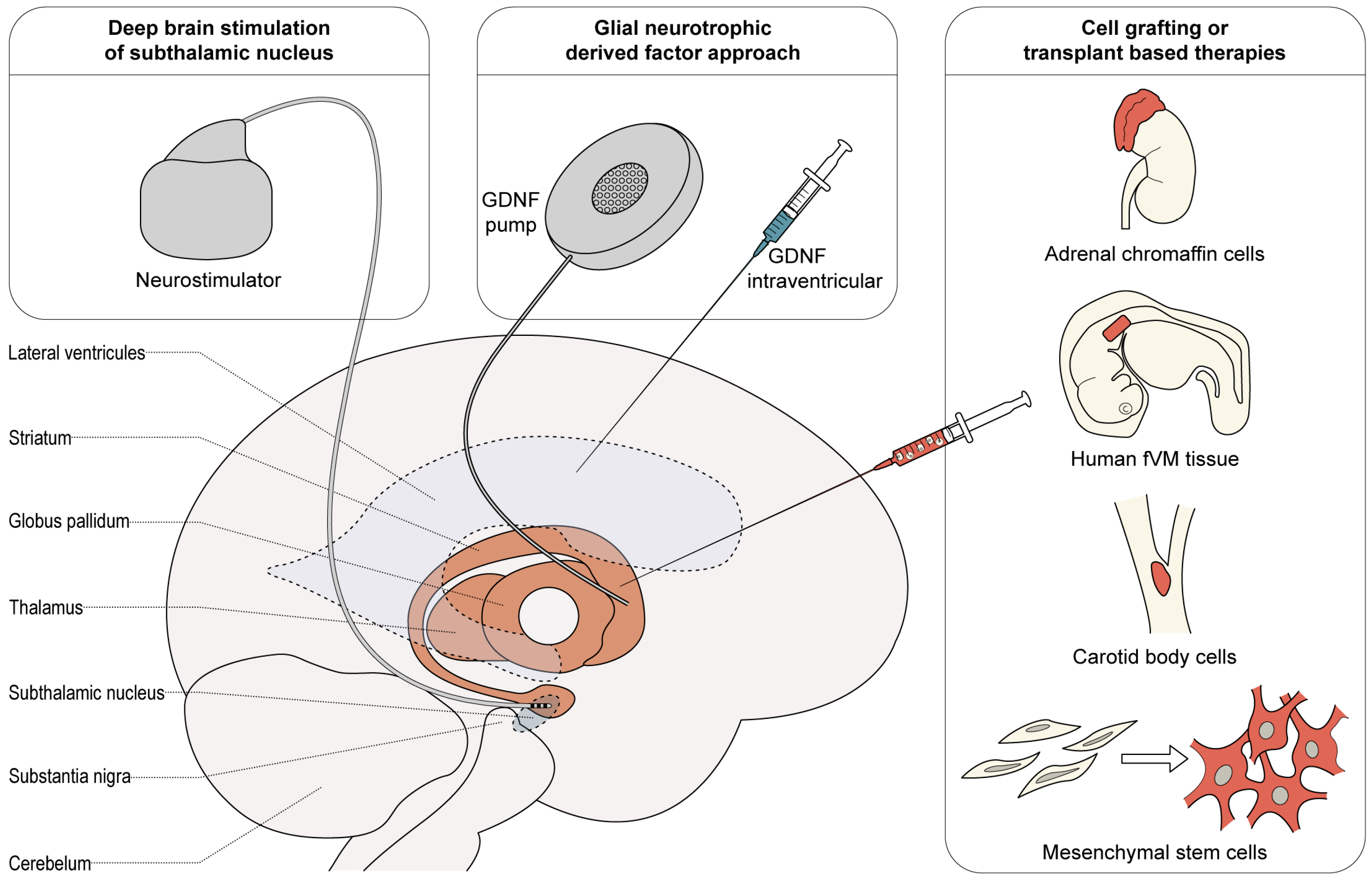
Neural Transplantation For Parkinson’s Disease: The Challenge
Despite continued research and development of drugs for the treatment of neurodegenerative diseases, disorders such as Parkinson’s disease , Huntington’s disease , and amyotrophic lateral sclerosis, are poorly treated. However, the degeneration and death of selected populations of neurons in these disorders-eg, dopaminergic neurons of the substantia nigra in PD-makes these diseases amenable to the development of cell-replacement therapies.
In the July issue of The Lancet Neurology, many aspects of cell-replacement therapies are described in a Forum article with contribution from leading investigators from Europe and the USA. The authors discuss the ethics of transplantation for PD, alternative cell sources, and how we should proceed with clinical trials. “Replacement of dopaminergic neurons in patients with Parkinson’s disease has spearheaded the development of this approach and was the first transplantation therapy to be tested in the clinic”, say Anders Bjorklund and Steve Dunnett in their introduction to the topic. “In our opinion, transplantation of fetal dopaminergic neurons is based on a solid rationale and open-label trials have provided convincing proof of principle that transplants can work-and work well-in some patients.”
The forum is broken down into six sections:
Neural transplants in Parkinson’s disease: do they work?Anders Bjorklund and Stephen B Dunnett
Are stem cells a valid alternative to human fetal tissue?Lorenz Studer
Gmp Cryopreservation Of Cells
The generation of good manufacturing practice -compliant, deliverable midbrain DA progenitors/neurons optimized for cell-based therapy for PD is a major challenge. Currently, a diverse collection of clinical-grade hESC lines are available as starting material to generate GMP-compliant mDA progenitors/neurons. In fact, GMP compliant differentiation protocols and reagents have been successfully applied to generate GMP mDA neurons .
Also Check: Mucuna Plants For Parkinson’s Disease
What Is Parkinson’s Disease
Parkinson’s Disease is a degenerative nervous system condition that affects one’s movement. Symptoms often start quite gradually, with minor issues such as small tremors within the extremities . Currently, there is no cure for Parkinson’s Disease but certain medications do have the capacity to help manage symptoms. Some doctors may also recommend surgery to address certain symptoms, which involves regulating certain areas of the brain.
Assessment Of The Efficacy Of Cell Transplants With Immunostaining Characterization
In the case of mDA progenitor neuron specifications, positive gene expression of common transcription factors FOXA2, LMX1A, and OTX2 and negative markers such as Afp, Gata4, and Brachyury have been quantitatively analyzed . More importantly, the upregulation and downregulation of these markers at a given stage in vitro governs the efficiency of cell fate determination. Unfortunately, these markers have been shown to coexpress in the diencephalic progenitor cells of the subthalamic nucleus . Furthermore, the expression of the positive genetic marker for DA neurons, tyrosine hydroxylase , a rate-limiting enzyme in dopamine synthesis , and the levels of GIRK2 have also been observed in many cell types in vitro . Moreover, common positive markers used to isolate high-quality DA progenitor cells include EN1 and SPRY1 Nurr1 FOXA2, LMX1B, and MSX1 , and the bicoid-related homeodomain factor Ptx3/Pitx3 . It is noteworthy that some discrepancies have been found with the requirement for the presence of floor plate-specific cell surface marker CORIN expression . A more recent study has identified a cell surface marker integrin-associated protein as a positive marker for FOXA2-positive DA progenitor cells .
Also Check: Does Parkinson’s Cause Neuropathy
The Next Generation Of Trials
Studer was part of the initial studies involving fetal tissue in the 1980s and 1990s, and knew from the start that the work was more of a proof of principle than a solution for people with Parkinsons. For me it was clear that a fetal transplant isnt a long-term solution because of ethical, legal and practical issues. Because this procedure requires 4 to 12 fetuses per patient, there was no way they could treat thousands, let alone tens of thousands, of people that way. Instead, Studer turned to stem cells.
Immunosuppression is a particularly important element of BlueRocks approach, because it relies on a single cell line that cannot be adjusted to more closely resemble the recipients own tissues. A group led by stem-cell scientist and neurosurgeon Jun Takahashi at Kyoto University in Japan is attempting to provoke a lesser immune response by pairing transplant recipients with cells that are less likely to be rejected. The researchers are using cell-surface proteins, called major histocompatibility complexes , that are recognized by the adaptive immune system and can have varying levels of compatibility from one person to another. Rather than using frozen cell lines, Takahashi and his colleagues are creating a fresh batch of MHC-matched cells for each transplant.
Research Progress Of Ipscs In The Treatment Of Pd
As early as 2009, CHAMBERS et al. obtained the midbrain DN derived from hiPSCs and confirmed that its function is similar to the DN derived from hESCs. To prepare a transplantable source of mDA cells, the key is to determine the best stage of cell differentiation in vitro. The optimal stage of transplantation of DN derived from hESCs has been fully studied.
DOI et al. tested the survival rate and function of CORIN sorted DN progenitor cells derived from iPSCs from healthy individuals or PD patients, and found that DN progenitor cells can improve the motor dysfunction in preclinical 6-OHDA-deficient rat models.
In 2018, Nature reported that the worlds first clinical trial of iPSCs for the treatment of PD was approved. Takahashi and his team transplanted neural precursor cells cultured with iPSCs into the brains of PD patients, with the hope that the transplanted cells will develop into neurons and be released Dopamine, and will test the effectiveness and safety of iPSCs in the treatment of PD.
Don’t Miss: Medical Alert Bracelets For Parkinson’s Disease