People Who Already Have Pd: Should I Get Tested And What Do I Do With The Results
Up until recently, even people with PD with a very extensive family history of PD would not necessarily receive genetic testing because there were no clear uses for the results. There has been research directed at figuring out whether PD caused by or associated with certain mutations have particular clinical characteristics . However, there remains so much variability in clinical characteristics even among people with the same PD mutation, that there are still no clear practical implications in knowing whether a PD patient harbors a particular mutation. There is also, so far, no difference in treatment or management of PD whether or not the patient harbors one of the known mutations. That may change however, with the advent of clinical trials that target particular mutations.
There are two genes that have received particular attention recently because medications are being developed that target those with mutations of these genes.
GBAis a gene that increases the risk of developing PD. The gene encodes for the GBA enzyme, a protein used by the body to break down cellular products. Having two abnormal GBA genes causes Gauchers disease, which is characterized by the buildup of these cellular products resulting in fatigue, bone pain, easy bleeding and an enlarged spleen and liver. When a person inherits only one abnormal gene, he or she does not develop Gauchers disease, but does incur a small increased risk of PD. Most people with one mutated GBA gene do not develop PD.
How Do Lrrk2 Gene Mutations Cause Parkinsons Disease
Healthy Individuals withoutLRRK2 Parkinsonâs disease
All humans have two copies of theLRRK2 gene which produce LRRK2 protein. In healthy people both copies of the gene are normal and therefore produce normal LRRK2 which regulates normal cellular biology.
Patients withLRRK2 Parkinsonâs disease
Patients withLRRK2 Parkinsonâs disease usually have one normal gene and one mutated gene. This means that patients produce both healthy LRRK2 protein and an overactive form of LRRK2 protein. The overactive LRRK2 is believed to drive neurodegeneration, causing Parkinsonâs disease, while the healthy LRRK2 continues to provide normal regulation to cells in other organs such as the lungs and kidneys.
Patients withLRRK2 Parkinsonâs disease â Non specific inhibition
In order to slow or stop the disease, ESCAPE Bio believes that overactive LRRK2 needs to be nearly fully inhibited. However, if healthy LRRK2 is also inhibited by the same amount then it may not be able to regulate cellular function in other organs, thereby driving histopathological changes in the lung and kidney, which are hard to detect and may be associated with irreversible pathology if they go unchecked.
Patients withLRRK2 Parkinsonâs disease â ESCAPEâs approach
Genetic Predispositions Reducing Differentiation Yield Of Mda Neurons
In vitro neural development was impaired in neural lines derived from patients carrying LRRK2, PRKN, SNCA, and sporadic mutations,,,. In four independent studies, the differentiation potential of neural progenitor cells derived from patients was significantly reduced, demonstrated by low yields of neurons in comparison with control lines,,,,. A recent review presented the idea that PD is attributed to significant neurodevelopmental defects, which may increase the susceptibility for disease onset. If confirmed, identifying genetic predispositions that contribute to early developmental defects in iPSC-PD may assist the development of novel PD therapies. However, these phenotypes may appear in conflict with other studies,, capable of generating functional neurons from cell lines with similar mutations. The differences could be due to varying protocols, which may be more or less stressful for the cells.
You May Like: Foods To Avoid With Parkinson’s Disease
Concluding Remarks And Future
Genes implicated in Mendelian forms of PD have provided new insights into the pathogenesis of the disease. The molecular pathways identified in monogenic cases may also be implicated in sporadic PD. The effect of dosage of SNCA on the phenotype of patients with duplications or triplications is illustrative. In addition, non-coding variants in this gene, thought to affect the level of expression in neurons, are associated with risk of the disease. The molecular mechanisms that contribute to PD and related disorders result in the death of dopaminergic neurons in vulnerable brain regions, and consequently the shared phenotype. However, known PD-causing genes account for only a small fraction of monogenic forms. Robust high-density SNP genotyping technologies and data analysis programs, combined with the analysis of copy number variations and large pathogenic genomic rearrangements, will identify novel loci. The clinical heterogeneity of parkinsonism is probably the cumulative effect of different gene-environment and/or genegene interactions. To identify risk variants in PD, association study methodology must be improved. Studies in isolated and heterogeneous populations, and approaches that minimize population stratification, are needed. Large-scale studies and publicly available GWA databases, crucial for statistical power, require collaborative efforts with shared sets of stringent clinical, genetic and analytic methods.
Common Symptoms Of Parkinsons Disease
Here are some of the early signs and symptoms of Parkinsons disease that you need to be on the lookout for in deciding to see a healthcare professional who can provide medical advice.
- Rigid muscles
- Lack of ability to write
- Slowed movement
- Low blood pressure
If you notice these, or other symptoms involving the nervous system, it is time to see a doctor. If you experience a sudden drop in the bodys ability to execute any of these tasks or control movements, it is a sign that something is wrong. Doctors may be able to give medications or other treatments that could improve symptoms.
Don’t Miss: Speech Therapy For Parkinson’s Disease
How Environmental Factors Could Cause Parkinsons Disease
Scientists differ about the extent that brain cells are impacted by environmental factors. However, the statistics associated with the disease show that the environment can play a very large role in whether parkinsons disease develops.
Most often, it is exposure to toxic chemicals that could play a role in the development of Parkinsons disease. Usually, these combine with genetic factors to produce the conditions that cause Parkinsons.
Increasing scientific evidence suggests that Parkinsons may be caused by environmental factors such as exposure to herbicides such as Paraquat.
How You Lose Dopamine Production
Damaged nerve cells can be what results in a decreased ability of the brain to create dopamine. Generally, some kind of degradation of the brain cells will reduce dopamine production.
There is some genetic link to parkinsons disease. For example, specific genetic mutations can impact the dopamine production. Far more common is that exposure to something in the environment can impact the brain.
Recommended Reading: Parkinson’s Disease Brain Changes
Predicting Risk Progression And Defining Etiological Subtypes Of Disease
Individually GWA identified loci confer relatively small amounts of disease risk however, the use of polygenic risk scores affords the ability to attribute a total known genetic risk score to an individual by summing their collective genetic risk. To date, the PRS reveals that, collectively, the 90 susceptibility loci confer considerable risk for disease, with those in the top decile of genetic risk being 6-fold more likely to have PD than those in the lowest decile of genetic risk . Additionally, by creating a composite risk score for PD diagnosis that combines the cumulative effect of genetic risk variants as well as the presence or absence of anosmia, age, sex, and family history, the ability to predict individuals at high risk for PD is remarkable, showing an AUC sensitivity of ~ 83.4% and specificity of ~ 90% .
Research focused on age at onset disease modifiers is one area where consistent effort has been seen. The largest PD age at onset GWAS to date included data from > 25K cases and identified two GWAS significant signals one at SNCA and the other was a protein-coding variant in TMEM175, both of which are known PD risk loci. Notably, these results showed that not all PD risk loci influence age at onset and therefore suggest the idea that risk and onset might operate through mechanisms that do not completely overlap.
When To See A Doctor About Parkinsons
There isnt one specific test to diagnose Parkinsons disease. Doctors will usually evaluate your symptoms and perform several tests to determine if you have the condition. If you notice the following early warning signs, then you should see a doctor.
The early warning signs of Parkinsons disease include:
Recommended Reading: Reishi Mushroom Parkinson’s Disease
Some Patients May Not Be Eligible To Take Part In The Free Genetic Testing Program
To speed up recruitment for a natural history program we are first focusing on patients who have a higher likelihood of testing positive for the mutated gene from the free genetic testing program. If a patient does not meet the criteria now, we will offer to contact them again should we open up the screening program to a wider population of Parkinsonâs patients.
What Should You Know About At
Genetic tests are not a substitute for a Parkinson’s diagnosis. Most at-home genetic tests do not provide genetic counseling services to help interpret the results. Always consult with a genetic counselor and your doctor before and after taking a genetic test. Most at-home genetic tests check for a limited number of gene mutations associated with Parkinson’s. This can be misleading since these tests may not be comprehensive.
Since scientists are still discovering more PD-associated genes, it is important to consult your doctor about comprehensive genetic testing options, like the PD GENEration study, which provides a free comprehensive genetic test identifying all possible variants in the two most common PD genes.
Even if you or a loved-one test positive for a Parkinson’s gene, it does not mean either of you will develop it. Having a genetic mutation only means that you may be at increased risk to develop PD. Environmental factors and lifestyle choices will help determine whether someone will get Parkinson’s.
Also Check: Apps For Parkinson’s Disease
Age And Genetic Factors Are Not Everything
The rate of Parkinsons disease globally has exceeded far faster than the population has aged according to the American Parkinson Disease Association.
Cases of the disease are up by several multiples over the past decades. From 1990 to 2015, the cases of the disease globally more than doubled, suggesting that there is far more at work. From 2015 to 2040, cases are expected to double once again. This is far higher than the rate of aging in the population.
Genetic Testing For Parkinsons Disease
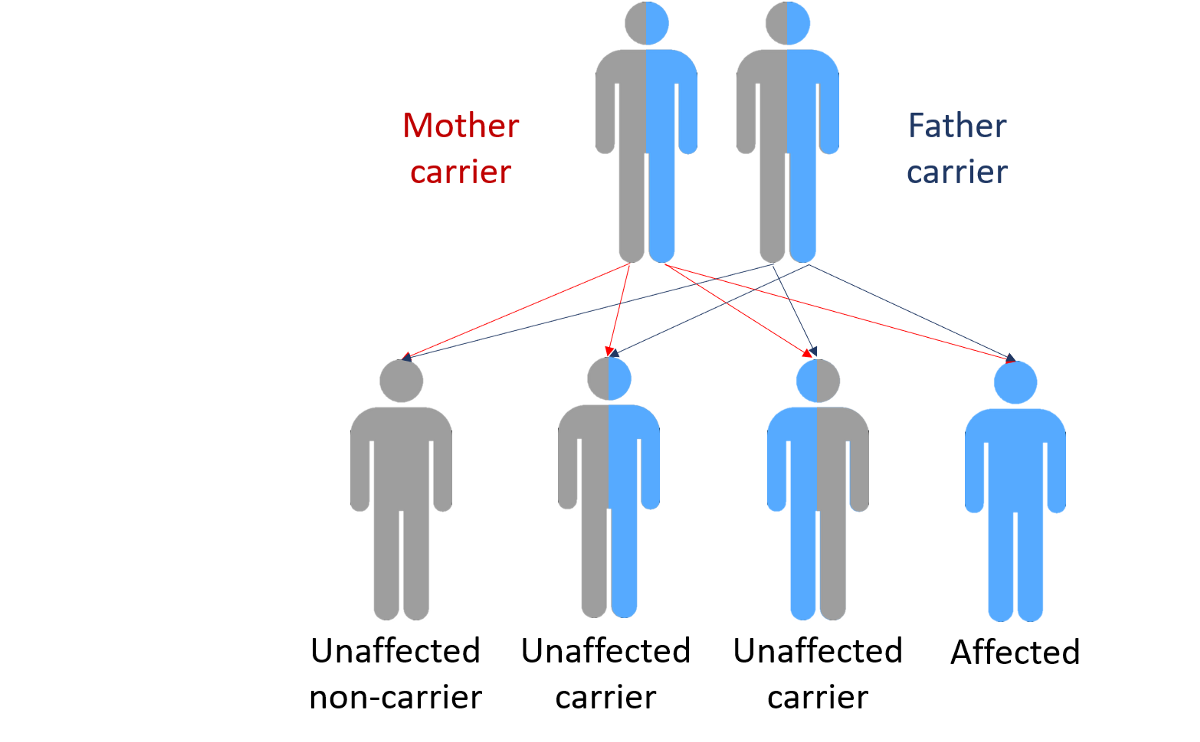
Similar to other complex diseases, the reason a particular person develops Parkinsons disease is likely a combination of genetic makeup and environment. In most people, the genetic contribution to disease development may be due to a number of different genes and the interactions between them. For only a very small percentage of people with PD, about 10%, the disease can be attributed to a single abnormal gene. Figuring out the identity and contributions of all the different genes that play a role in disease development is a very hot topic in PD research today.
Recommended Reading: How To Tell Difference Between Parkinson’s And Essential Tremor
The Interplay Between Genomic Predispositions And Environmental Factors Leads To Parkinsons
In the mid-1990s, the connection between PD and underlying genetic mutations was established,,. It is now evident that varying degrees of the interplay between genomic predispositions and aging and cellular stressors impose a risk for disease . Previous studies have shown vascular insults to the brain, repeated head trauma, neuroleptic drugs, exposure to pesticides, and manganese toxicity increase the risks of developing symptoms of PD,,. In addition, advancing age can also cause a cascade of stressors within the substantia nigra, which weakens the neurons and their ability to respond to further insults,. Ultimately, the uniqueness of the interactions between genes and the environment makes the development of a single treatment for PD difficult as they give rise to a spectrum of neuronal phenotypes that can be unique to individual patients . The development of a model with the ability to replicate the genomic and epigenetic aspects of the disease is crucial . As increasing evidence suggests that genetic mutations are key modulators of disease initiation and progression, the identification and understanding of the various genomic predispositions are required for the development of better-targeted treatments to slow the disease progression.
Fig. 1: A combinatorial spectrum of genetic risks, cellular stressors, and brain cell dysfunctions causes Parkinsons disease.
Reasons Why Parkinsons Disease Occurs
The scientific reason given for Parkinsons disease is that the patient has lost nerve cells in the part of the brain called the substantia nigra. A very important chemical called dopamine is produced by the substantia nigra. The loss of the ability to produce dopamine contributes to the early stages of Parkinsons disease.
You May Like: Parkinson’s Disease Advocacy Groups
Assessment Of Family History
The majority of our identified studies use self reporting or self administered questionnaires to assess family history of PD. Categorising PD cases who have relatives with isolated tremor as having a positive family history, can significantly increase the number of familial cases, especially among early onset PD cases.
Performing individual examinations may increase the precision with which a diagnosis of PD is made in relatives of cases and controls, rather than reliance on patient reporting of diagnoses or symptoms such as tremor. It has also been shown that significant numbers of previously unrecognised PD patients can be identified by examination despite a negative family history. It can often however be difficult verifying familial diagnoses in diseases affecting the elderly as relatives are often deceased and not subjected to postmortem examination. Subclinical Parkinson’s disease, diagnosed on the basis of Lewy body pathology in people without prior symptoms of PD, is observed in up to 10% of individuals subjected to postmortem neuropathological examination. No study includes pathological examination of all relatives of both cases and controls, which currently represents the gold standard in diagnosing PD.
Genetic Testing May Lead To A Cure
Although genetic testing can leave individuals with many unanswered questions, the data provided may further the study of the disease.
The more individuals you can work, the more things you can discover, says Cannon. We are interested in studying people who have a risk gene because the sooner we can learn how to stop it , the better off people will be.
Clinical trials are in progress to test therapies that target gene mutations, in particular GBA and LRRK2. Pharmaceutical companies conducting these studies need patients who test positive for specific gene variations. By getting tested, individuals have a chance to participate in research programs that may lead to a cure.
Gilbert points out that drugs that target specific mutations may benefit a larger group of Parkinsons patients.
The biochemical problem that happens when a person has an LRRK2 mutation might appear in someone else without an LRRK2 mutation but by another means, she says. So they may also benefit from medication developed for people with an LRRK2 mutation.
If you are interested in participating in a trial, the Michael J. Fox Foundation offers a roundup of the latest investigations currently being conducted and how to get involved.
Recommended Reading: Pre Parkinson’s Disease Symptoms
Will Findings From Pd Ipsc Models Translate To Human Clinical Trials
One of the most exciting applications of patient-derived iPSC models of PD is to validate pharmacological treatments before clinical trials. The field is still at the stage of improving human brain tissue engineering, and many different protocols are being tested and developed. However, the need for progress in clinical translation for brain disorders is extremely high, and there is no time to wait for brain tissue models to be perfect. Pioneering iPSC studies pave the road to success and identify limitations which help the community to reach a consensus on the minimal requirements to model brain disorders in vitro most accurately. It seems essential to improve the efficiency of reprogramming and differentiation protocols while trying to make those models as physiological and realistic as possible,. Some concerns are raised that in vitro neuronal development, maturation and function might be too artificial, suggesting that the model may overlook some of the critical processes that occur in vivo. Nevertheless, some defects observed in iPSC-derived neurons have already been confirmed in human postmortem brain tissues,,,,. Although this is very encouraging, it is unclear whether significant in vitro phenotypes that cannot be confirmed in postmortem brain tissue should be disregarded. Most postmortem brains also have technical limitations and may represent later stages of the disease, whereas iPSC models may represent earlier stages, preceding neurodegeneration.
Parkinson’s Disease Genetic Influence
Recent developments in research gene research has found that genetic influence plays a large role in Parkinson’s disease. Five main genes that are believed to contribute to the disease have been identified and located. These include alpha-synuclein, Parkin, Ubiquitin carboxyl-terminal hydrolase, DJ-1 and SCA2. It has been found that mutations of these genes are some of the underlying causes. In some causes there can be multiple mutations in one gene. The effects of some of these mutations are now understood.
Researchers suspect that genes associated with the late onset of Parkinson’s Disease are susceptibility genes rather than causal genes. It is thought that environmental factors act on these gene, consequently leading to Parkinson’s disease. But the mechanism in which they do so is not yet known. Researchers believe that if they can work out this mechanism, they can disrupt it in some way, and therefore halt the onset of the disease.
The general consensus among researchers is that both genetic influence and environmental factors lead to the onset of Parkinson’s Disease. The mechanisms of the genetic influence of Parkinson’s Disease are still to be understood and much more research is required.
The content of this web site is intended to convey general educational information and should not be relied upon as a substitute for professional healthcare advice. More information.
Recommended Reading: How Do You Treat Parkinson’s Disease Naturally