Exosomes Derived From Mesenchymal Stem Cells Repair A Parkinsons Disease Model By Inducing Autophagy
Journal: Cell Death and Disease
Institution: Hebei University of Chinese Medicine
Research Areas: Stem cell therapy
Cell Lines: hucMSC
Summary: Human umbilical cord mesenchymal stem cells can provide neuroprotection in neurodegenerative diseases, such as Parkinson’s disease, treatments. However, at the moment, hucMSC applications are limited due to various reasons. In this study, H.-X. Chen with colleagues has shown that Exosomes derived from hucMSC have the potential for effective Parkinson’s disease treatment because they can pass the Brain-Blood barrier. Here, HoloMonitor M4 was used to characterize hucMSC cell proliferation and helped to characterize hucMSC self-renewal and reproductive abilities.
Keywords: HoloMonitor M4, cell proliferation,mesenchymal stem cells
Modulatory Effects Of Mscs On Pd
Recently, dysregulation of the autophagy system has been identified in the brains of PD patients and animal models of the condition, suggesting a potential role for autophagy in PD . In PD models, MSCs have been demonstrated to improve a-syn clearance and regulate autophagy-lysosomal activity . MSCs may activate autophagy signaling through upregulation of Beclin-1 , a key positive regulator of mammalian autophagy. The secretome of MSCs has been found to contain numerous components associated with autophagy signaling in cell-based experiments through induction of autophagy-related genes, including beclin-1 , Gamma-aminobutyric acid receptor-associated protein-like 1 and Autophagy related 12 . The secretome of MSCs drives PI3K/Akt activation and modulates different signaling pathways to improve nutrient absorption, cell growth, metabolism, and proliferation .
According to several investigations, MSCs exhibit immunomodulatory effects after infiltrating to injury sites in response to particular chemotactic recruitment and releasing numerous growth and immunoregulatory factors, so they can alleviate inflammation and improve tissue healing . Therefore, MSC-based cell therapy has been used to modulate inflammation and accommodate tissue regeneration in treating many neuroinflammatory and neurodegenerative illnesses such as Parkinson’s disease .
Studies Show Promising Results
“Considering the ability of MSCs to secrete neurotrophic factors, modulate inflammation, and possibly even act as mitochondria âdonorâ, it comes as no surprise that there is a lot of interest in the use of MSCs in the treatment of Parkinsons Disease, and a multitude of animal studies has shown promise. Treatments have resulted in improvement of motor function, protection of the nigrostriatal system, and improved striatal dopamine release in several studies using toxic lesion rodent models of Parkinsons Disease. Similar effects were reported with umbilical cord-derived MSCs with or without prior differentiation. For example, a recent study reported improvement of motor function, reduced microglial activation, and decreased loss of TH immunoreactivity, associated with local production of trophic factors.
Also Check: Early Onset Parkinson’s Symptoms
Mscs And Their Differentiation Ability
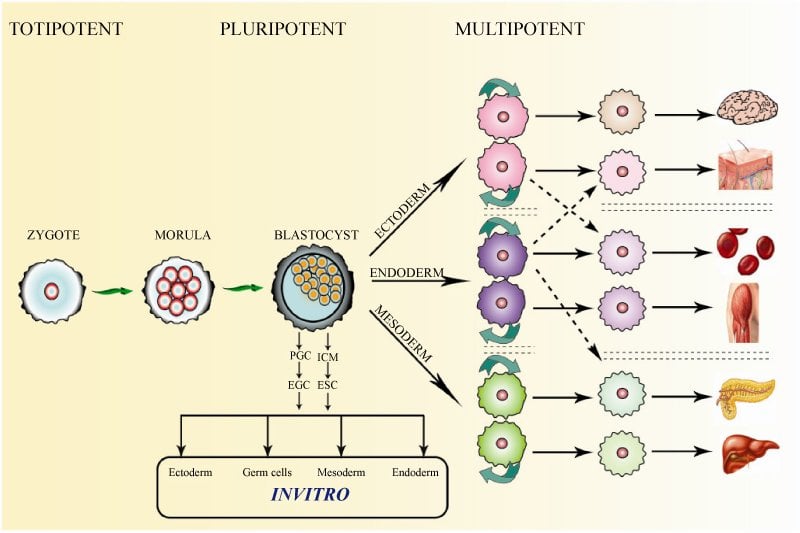
The possibility of MSC plasticity and transdifferentiation was initially described following in vivo experiments in which transplanted donor bone marrow-derived cells differentiated into glial cells in the recipient brain . While some studies suggested that MSCs are plastic based on their expression of cell-specific markers, the functions of the transdifferentiated cells were not clearly demonstrated in other cases. Moreover, questions have been raised regarding the interpretation of transdifferentiation of infused cells into neuronal lineage cells because some investigators have suggested that the transdifferentiation observed was rather a result of fusion between infused bone marrow cells and the host brain cells . Despite this uncertainty, accumulating evidence supports the broad differentiation of MSCs both in vivo and in vitro. Based on the frequency and ratio of MSCs integrated and differentiated into the host tissue, fusion alone cannot explain all of the phenomena observed after MSC infusion. Furthermore, experiments using a Cre-lox system clearly demonstrated that MSCs can transdifferentiate into epithelial cells in vivo without fusion . In vitro differentiation of MSCs provides further evidence for MSC transdifferentiation because there are no preexisting differentiated cells to be fused at the beginning of induction under culture conditions.
Human Umbilical Cord Mesenchymal Stem Cells Improve Locomotor Function In Parkinsons Disease Mouse Model Through Regulating Intestinal Microorganisms
- 1Department of Neurology, The First Hospital of Hebei Medical University, Shijiazhuang, China
- 2Hebei Provincial Engineering Laboratory of Plant Bioreactor Preparation Technology, Shijiazhuang, China
- 3Research Center, Hebei University of Chinese Medicine, Shijiazhuang, China
- 4College of Integrated Chinese and Western Medicine, Hebei University of Chinese Medicine, Shijiazhuang, China
- 5Affiliated Hospital of Hebei University of Engineering, Handan, China
Don’t Miss: Parkinson’s Disease And Tremors
What Are The Symptoms Of Parkinson’s Disease
Parkinson’s DIsease can include a variety of symptoms that vary in severity and type amongst the affected population. Early signs of the condition can sometimes go unnoticed but as the disease progresses one can expect these symptoms:
- Difficulty speaking
- Difficulty writing
- Loss of automatic movements
- Slowed overall movement
- Muscle stiffness
Pathophysiology Of The Disease
Basal ganglia motor circuitry: Parkinson’s disease is predominantly a disorder of the basal ganglia, which are a group of nuclei situated at the base of the forebrain . The striatum, composed of the caudate and putamen, is the largest nuclear complex of the basal ganglia. The striatum receives excitatory input from several areas of the cerebral cortex, as well as inhibitory and excitatory input from the dopaminergic cells of the substantia nigra pars compacta . These cortical and nigral inputs are received by the spiny projection neurons, which are of 2 types: those that project directly to the internal segment of the globus pallidus , the major output site of the basal ganglia and those that project to the external segment of the globus pallidus , establishing an indirect pathway to the GPi via the subthalamic nucleus . The actions of the direct and indirect pathways regulate the neuronal output from the GPi, which provides tonic inhibitory input to the thalamic nuclei that project to the primary and supplementary motor areas .
Figure 2: Anatomy of basal ganglia. View Figure 2
Two pathways exist within the basal ganglia circuit, the direct and indirect pathways , as follows:
Figure 3: Basal ganglia circuitry in Parkinson’s disease. View Figure 3
â¢In the direct pathway, outflow from the striatum directly inhibits the GPi and SNr striatal neurons containing D1 receptors constitute the direct pathway and project to the GPi/SNr
Common symptoms:
b.Terminates with movement
Don’t Miss: Parkinson’s And Extreme Fatigue
Preclinical Application Of Dpscs In Parkinsons Disease
However, one of the challenges is to find a safe and effective method of cell delivery before DPSC transplantation is used in clinical applications. It has been reported that the survival rate of transplanted cells, the full enrichment of therapeutic cells in the brain, and success in avoiding the distribution of stem cells to peripheral organs are all affected by transplantation methods . Due to the existence of the blood-brain barrier , DPSC transplantation is also faced with the problem of invasiveness and invalidity. However, some studies have explored the nasal system as a new pathway for stem cell delivery, which can bypass the BBB and directly target brain therapy for PD . Therefore, a recent study showed that degenerated tyrosine hydroxylase -positive neurons, motor coordination, and olfactory function were significantly improved by intranasal administration of PKH26 pre-labelled DPSCs into MPTP-induced PD mice . In another study, intrathecal injection also significantly promoted the recovery of neurons and the improvement of behavioural function . Comparatively, the method of intrathecal injection may be cumbersome and difficult. Regardless of how the DPSCs are administered, the ability of DPSCs to differentiate into DAergic neurons in vivo or in vitro and its positive role in PD therapy have been convincingly confirmed.
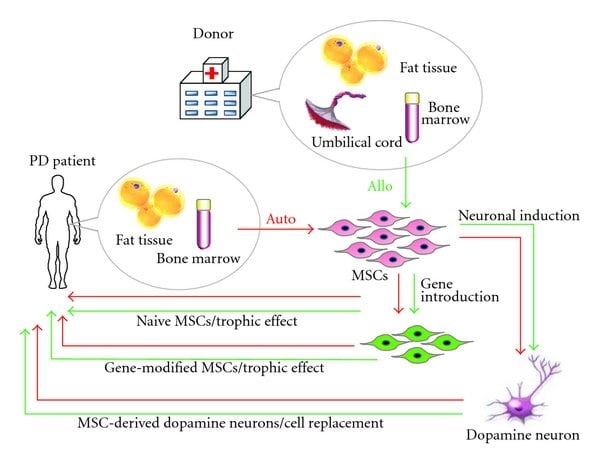
Mesenchymal Stem Cell Therapies For Neurodegenerative Diseases
While there have been significant advances in the symptomatic management of these diseases that improve quality of life and at times survival, the available medications likely only slow the progression of neuronal death by a few months. The idea of using cell therapy to treat neurodegenerative diseases has been around for decades, most notably in Parkinson’s Disease where a variety of cell transplant investigations have been performed with success.
According to a recent study conducted by Nathan P. Staff et al,
“The precise mechanism by which MSCs may exert beneficial effects in neurological disease is still being elucidated, but it appears that multiple different mechanisms may contribute. First, MSCs have been shown to secrete neurotrophic growth factors, including glial cell-derived neurotrophic factor , vascular endothelial growth factor, and brain-derived neurotrophic factor ,which can be further enhanced under specific culture conditions.Neurotrophic growth factors have been shown to improve neuronal survival in a number of preclinical models of neuron injury, including ALS, PD, and MSA transgenic animalsand nerve injury models. â Second, MSCs strongly modulate the immune system and can aid wound healing, and this mechanism has been exploited in disorders such as graft versus host disease and Crohnâs disease. From a neurodegenerative perspective, it has become increasingly recognized that neuroinflammation plays a significant pathomechanistic role.”
Also Check: What Are The Signs Of Parkinson’s Disease
Preclinical Application Of Shed In Parkinsons Disease
Previous studies on the neural potential of DPSCs and SHED without neural induction have shown that these cells can express nestin, a neural progenitor cell marker, and GFAP, a glial cell marker, at the mRNA and protein levels . In vitro differentiation studies have also shown that they can differentiate into nerve cells and can survive and express neuronal markers when SHED is transplanted into the brain of adult rodents. In addition, they are neural crest-derived cells and are easy to obtain, which further indicates their potential application value for the treatment of PD.
Based on the above studies, there may be three main mechanisms of SHED in the treatment of PD . First, DAergic neurons or other neurons differentiated by SHED in vivo form a functional connection with the host neuron. Transplanted DAergic neuron-like cells can secrete DA to restore the functional activity of neurons. Second, the transplanted SHED can secrete cell growth factors, such as VEGF, BDNF, and GDNF. Some studies have reported that undifferentiated MSCs can secrete neurotrophic factors such as BDNF, GDNF, NGF, HGF, and VEGF to play a neuroprotective role . Third, through the immune regulation by cytokines such as IL-6 and TNF-, these factors may combine with VEGF, BDNF, and GDNF to enhance immune regulation and reverse the damage to host neurons .
What Causes Parkinson’s Disease
Parkinson’s Disease is caused by a loss of nerve cells in the brain. This loss of nerve cells within the brain results in a reduced amount of dopamine being created which acts as a messenger between the parts of your brain that control voluntary and involuntary movement. Therefore without that vital connection, your brain starts losing the ability to effectively control movement. Currently, it is unknown what causes the deterioration of nerve cells associated with Parkinson’s Disease . Currently, it is believed that both environmental factors, as well as genetic factors, may play a role in the loss of nerve cells.
Parkinson’s Disease is a lifelong condition that can greatly impair the ability of one’s daily functions. Traditional treatments only address the symptoms of the condition, but researchers are excited about the possibilities of certain gene therapies and stem cell therapy, which may have the ability to reverse damage and halt the progression of the disease.
You May Like: Is Parkinson’s Disease Hereditary
Induction Of Functional Dopamine Neurons From Bmscs
A system to specifically induce dopamine neurons from BMSCs was reported . This system first generates postmitotic functional neuronal cells with a very high efficiency without contamination by glial cells. The resulting neuronal cells are then further induced into dopamine neurons. The induction is achieved by lipofection of a plasmid containing a Notch1 intracellular domain and G418 selection, followed by the administration of a specific combination of trophic factors and cytokines .
Induction of dopamine neurons from MSCs. After NICD introduction, MSCs become similar to NPCs, expressing the NPC markers nestin, GLAST, 3-PDGH, and neuroD. After cytokine stimulation ), cells become postmitotic neurons expressing neuronal markers such as neurofilament, Tuj-1, and MAP-2. The administration of GDNF induces neurons to become dopamine neurons , which are useful in the Parkinson’s disease model. Pictures from J Clin Invest 113 17011710 and J Cereb Blood Flow Metab 29 14091420 .
When NICD-introduced BMSCs are expanded and then stimulated with trophic factors ) for several days, approximately 96% of the cells extend neurites and differentiate into postmitotic neuronal cells. These cells are positive for the neuronal markers MAP-2ab, neurofilament, and Tuj1, and most importantly, action potentials were recorded in the cells in a patch clamp experiment, suggesting that these induced cells are functional neuronal cells .
Phase Iia Randomized Placebo Controlled Trial: Mesenchymal Stem Cells As A Disease
| The safety and scientific validity of this study is the responsibility of the study sponsor and investigators. Listing a study does not mean it has been evaluated by the U.S. Federal Government. Read our disclaimer for details. |
| Recruitment Status : Active, not recruitingFirst Posted : August 10, 2020Last Update Posted : February 10, 2022 |
- Study Details
| Drug: MSC+placeboDrug: MSCDrug: Placebo | Phase 2 |
| Study Type : | |
| Triple | |
| Primary Purpose: | Treatment |
| Official Title: | A Randomized, Double-blind, Placebo-controlled Trial of Allogeneic Bone Marrow-derived Mesenchymal Stem Cells as a Disease-modifying Therapy for Idiopathic Parkinson’s Disease |
| Actual Study Start Date : |
| 2 treatment doses + 1 placebo 3 months apart | Drug: MSC+placebo2 infusions of 10 X 10^6 MSC/kg and 1 placebo every 3 months.Other Name: allogeneic mesenchymal stem cell or similar placebo |
| Experimental: MSC3 treatment doses 3 months apart | Drug: MSC3 infusions of 10 X 10^6 MSC/kg every 3 months.Other Name: allogeneic mesenchymal stem cell |
| Placebo Comparator: Placebo3 placebo doses 3 months apart | Drug: Placebo3 infusions of placebo every 3 months. Placebo will be identical to the investigational product but will not contain MSCs.Other Name: Similar placebo |
Also Check: How To Manage Parkinson’s
Mptp Injury Mouse Model And Uc
Sixty male C57BL/6 mice were purchased from Vital River Laboratory Animal Technology Co., Ltd. . The mice were housed in a specific pathogen-free laboratory under a controlled environment with a temperature of 22 ± 3°C and humidity 60 ± 5% at 12-h light/12-h dark cycle. All mice were given free access to food and tap water. The experimental protocols and animal care were strictly in accordance with the approval of the Animal Care and Management Committee of Hebei Medical University.
The mice were randomly assigned to receive either intraperitoneal injection of MPTP or normal saline. The MPTP-induced PD mouse model was conducted as previously described . MPTP was injected intraperitoneally once a day for 5 days to produce an experimental PD model.
The method of UC-MSC administration is modified according to previously published . Intranasal application of UC-MSCs or PBS into MPTP-or vehicle-treated mice was performed 5 days after MPTP injection. Two hours after MPTP injection, each nostril was treated with 5.0 l of hyaluronidase in sterile PBS solution to enhance the permeability of the nasal mucous membrane. Thirty minutes later, 5.0 l cell suspension was instilled in the nasal cavity with a pipette in a 5-min interval. The daily dose contained 1 × 106 cells/40 l .
What Are Researchers Investigating Can Stem Cell Therapy Improve The Symptoms Of Parkinsons Disease
Frustration from limited treatment options has led to an increased focus on stem cell replacement therapy, treatment intending to provide long-lasting relief from symptoms of Parkinsons disease.
Approximately 5% of patients diagnosed with Parkinsons disease have an inherited genetic mutation.
Researchers are currently investigating the underlying cause of Parkinsons disease in the other 95% of cases.
When dopamine was reintroduced into the central nervous system, symptoms were decreased or reversed. This means, if stem cells can be induced into becoming dopamine producing neurons and then transplanted in affected zones, they could replace impaired cells, improving function.1,2
Researchers have shown transplanting young brain cells derived from human umbilical cord into patients with Parkinsons disease resulted in an improvement in disease related symptoms.
Animal studies have shown mesenchymal stem cells promote neuroprotection and neurodifferentiation, by modulating neural stem cells, neurons and glial cells and axonal growth.3-11 They have the ability to repair and regenerate neurons in the brain, reduce levels of free radicals, improve synaptic connection from damaged neurons and regulate inflammation.
Animal models have shown stem cell therapy has been shown to be safe and effective.
Read Also: How To Care For Someone With Parkinson’s Disease
Measurement Of Cytokines In Serum And Colon
Blood was collected via the orbital venous plexus with anticoagulant-free tubes. Blood was centrifuged at 4500 g for 10 min at 15°C, and serum was isolated and stored at 80°C until it was used. The contents of tumor necrosis factor-alpha and interleukin 6 in serum and the colon were measured using ELISA kits according to the protocol of the manufacturer. The contents of lipopolysaccharides in serum were measured with commercial kits according to the manufacturers instructions.
Cell Culture And Phenotype Identification
Fresh umbilical cord samples were obtained from normal spontaneous full-term delivery mothers with written informed consent and reserved in a sterilized phosphate-buffered saline solution processed within 3 h. The cord was rinsed three times to remove the residue blood and clots, cut into 3-cm-long pieces, and rinsed again in a petri dish until the solution became clear. After blood vessels were removed, Whartons jelly was dissected into pieces approximately 0.3 cm3 in size and then transferred into culture vessels, with 10 ml mesenchymal stem cell complete medium at 37°C in a 5% CO2 incubator. The medium was replaced with fresh medium every 3 days after the initial plating. The cultured cells were passaged when cell confluency reached 80%.
Don’t Miss: What Kind Of Doctor Treats Parkinson’s Disease